In post hoc and real-world analyses, SARCLISA-based regimens demonstrated efficacy1,2
Across cytogenetic risk profiles
Regardless of early or late relapse
Regardless of the number of prior lines of therapy
Cytogenetic Risk Profiles
SUBGROUP ANALYSIS OF THE ICARIA-MM TRIAL
SARCLISA + Pd showed 19.1 months mPFS and tripled ≥CR in the standard-risk subgroup and doubled PFS in ultra-high-risk subgroup1
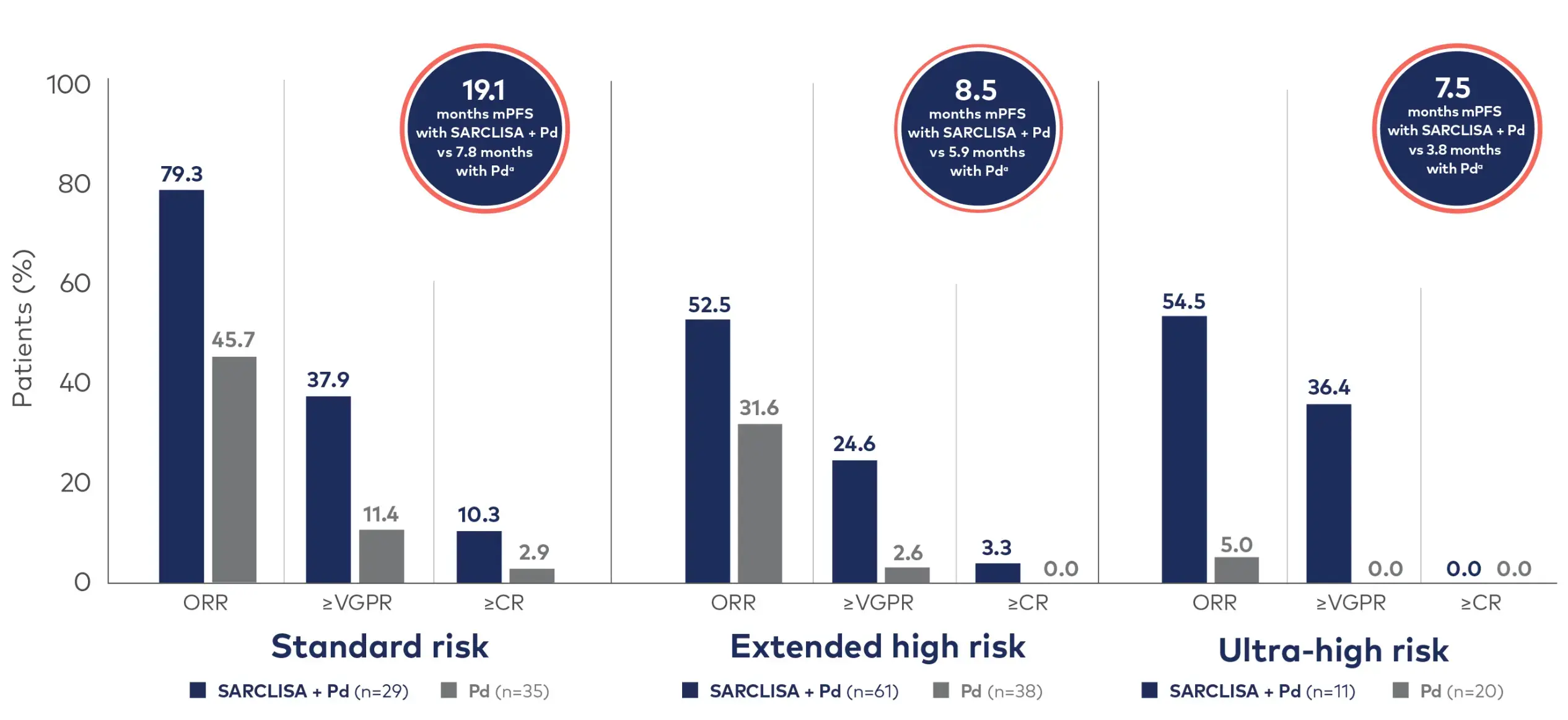
- Standard risk was defined as the absence of the following high-risk cytogenetic abnormalities: del(17p), t(4;14), t(14;16), and 1q21+, which included both gain(1q21) and amp(1q21)1
- Extended high risk was defined as the presence of 1 of these high-risk cytogenetic abnormalities1
- Ultra-high risk was defined as the presence of ≥2 high-risk cytogenetic abnormalities1
mPFS was measured for different subgroups, including standard-risk at 19.1 months (HR=0.478 [95% Cl: 0.263, 0.866]), extended high-risk at 8.5 months (HR=0.645 [95% Cl: 0.414, 1.006]), and ultra-high-risk at 7.5 months (HR=0.437 [95% Cl: 0.188, 1.018]).1
OS rates were measured for standard-risk (HR=1.032 [95% CI: 0.542, 1.972]), extended high-risk (HR=0.842 [95% CI: 0.533, 1.330]), and ultra-high-risk subgroups (HR=0.796 [95% CI: 0.357, 1.776]).1
Key takeaway
SARCLISA + Pd showed consistent efficacy across cytogenetic risk profiles and real-world effectiveness regardless of the number of lines of prior therapies1,3
For more information about the phase 3 ICARIA-MM study, view SARCLISA + Pd trial design.
ICARIA-MM Safety Profile1
SARCLISA + Pd was well tolerated across all risk category subgroups in the phase 3 ICARIA-MM trial. The incidence of Grade ≥3 treatment-emergent adverse events was generally higher in the SARCLISA + Pd arm than the control arm, regardless of risk profile.1
In the ICARIA-MM dataset, standard-risk patients with the most frequent Grade ≥3 TEAEs and haematologic abnormalities were neutropenia (82.8%), thrombocytopenia (31.0%), anaemia (27.6%), and pneumonia (24.1%), with fewer cases of infusion-related reactions and hypertension (3.4% each), and no cardiac failure. Extended high-risk patients reported neutropenia (88.3%), thrombocytopenia (31.7%), anaemia (31.7%), and pneumonia (25.0%), with no infusion-related reactions or cardiac failure and lower rates of hypertension (1.7%). Ultra-high-risk patients showed neutropenia (81.8%), thrombocytopenia (54.5%), anaemia (36.4%), pneumonia (27.3%), and infusion-related reactions (9.1%), with no hypertension or cardiac failure.1
Number of Prior Lines of Therapy
REAL-WORLD EVIDENCE
mPFS was not yet reached in SARCLISA + Pd patients who received 1 line of prior therapy3
The IMAGE study,* a retrospective cohort analysis, evaluated the real-world effectiveness of SARCLISA + Pd in French EAP-treated patients.3
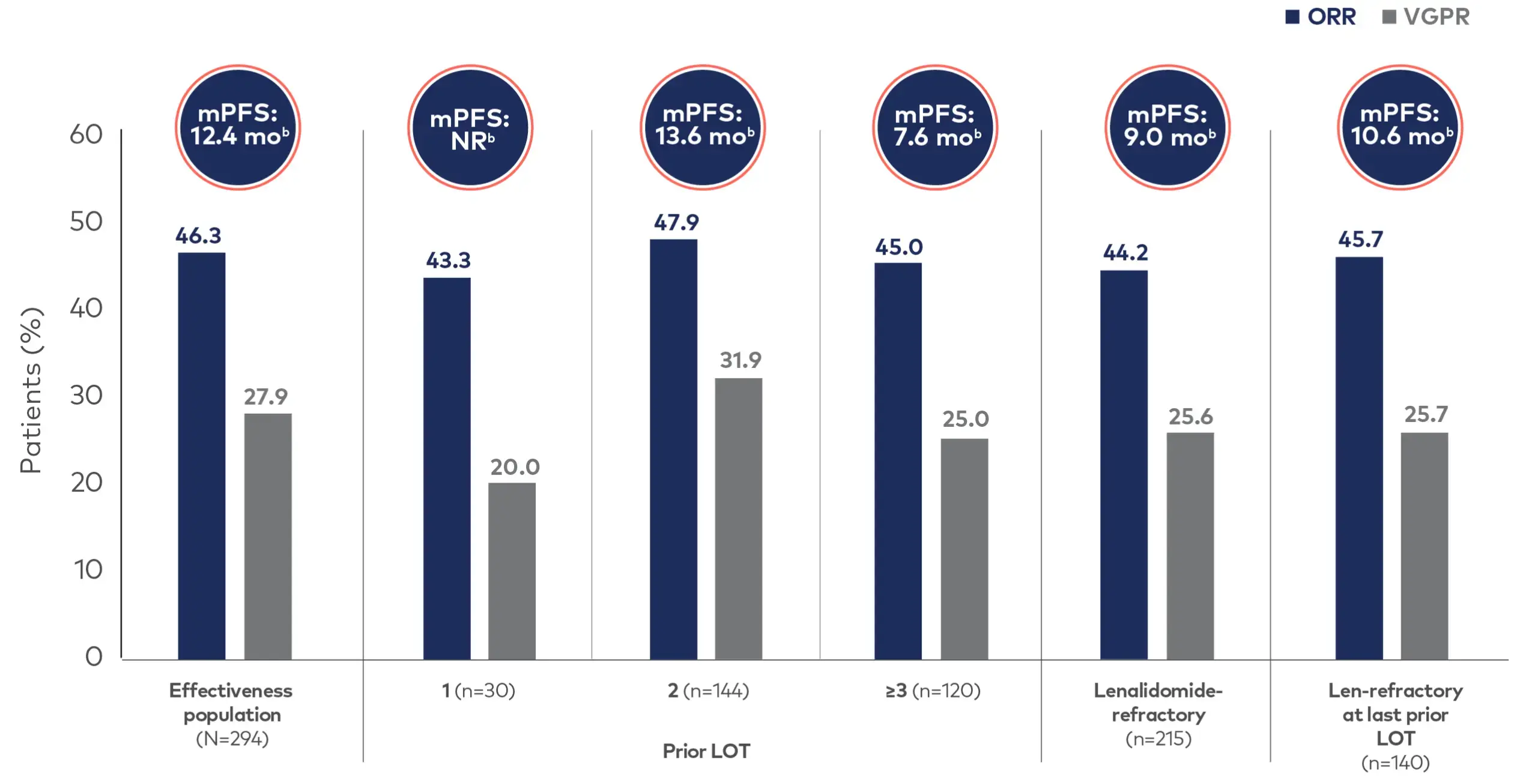
Adapted from Decaux et al. Eur J Haematol. 2024. doi:10.1111/ejh.14225. See original graphics in Decaux et al. (Figure 3).
For the anti-CD38–naïve population (n=216), mPFS, ORR, and ≥VGPR were 16.6 months (95% CI: 13.2, NR), 50.0%, and 31.5%, respectively.3
Median OS was NR (95% CI: 18.9, NR) in the IMAGE study.3
Key takeaway
In a real-world setting, SARCLISA + Pd showed effectiveness and depth of response in patients who received 1 prior line of therapy or more, consistent with that in the overall effectiveness population.3
For more information about the phase 3 ICARIA-MM study, view SARCLISA + Pd trial design.
IMAGE Study Safety Profile3
No new safety signals were observed in the IMAGE study compared with ICARIA-MM. However, the incidence of some AEs like infections was low, possibly due to the retrospective nature of the study.3
In the safety population (n=299), 79 patients (26.4%) reported at least one adverse event. There were no new safety concerns identified, and no cases of viral re-infection were reported among the patients. One patient (0.3%) was noted to have developed a secondary primary malignancy. Only 4 patients (1.3%) permanently discontinued SARCLISA due to an AE, and there were no cases where SARCLISA was permanently discontinued due to neutropenia.3
aThe 95% confidence interval for standard risk, extended high risk, and ultra-high risk were (0.263, 0.866), (0.414, 1.006), and (0.188, 1.018), respectively.1
bThe 95% confidence interval for the effectiveness population was (9.0, 15.0). For those receiving 1, 2, or ≤3 prior LOT it was (9.6, NR), (10.2, NR), and (4.4, 11.8), respectively. For lenalidomide-refractory patients (6.4, 12.4) and those who are refractory at last prior LOT, (14.8, NR).3
*IMAGE: a retrospective cohort study conducted in France among patients enrolled in EAPs for RRMM. It evaluated the safety and efficacy of SARCLISA + Pd in 294 patients. PFS, response rates, and coded adverse events were key endpoints.3
AE=adverse event; CR=complete response; EAP=early access program; HR=hazard ratio; IRC=Independent Review Committee; LOT=line of therapy; mPFS=median progression-free survival; NR=not reached; ORR=overall response rate; OS=overall survival; Pd=pomalidomide and dexamethasone; PFS=progression-free survival; RRMM=relapsed and/or refractory multiple myeloma; TEAE=treatment-emergent adverse event; VGPR=very good partial response.
References: 1. Moreau P, Perrot A, Dimopoulos MA, et al. Isatuximab in relapsed multiple myeloma patients with ultra-high-risk cytogenetics: ICARIA-MM and IKEMA subgroup analysis. Poster presented at: European Hematology Association (EHA) 2023 Hybrid Congress; June 8-11, 2023; Frankfurt, Germany and online. 2. Facon T, Moreau P, Baker R, et al. Isatuximab plus carfilzomib and dexamethasone in pts with early versus late relapsed multiple myeloma: IKEMA subgroup analysis. Presented at: 64th ASH Annual Meeting and Exposition; December 10-13, 2022; New Orleans, LA. ID #753. doi:10.1111/ejh.14225 3. Decaux O, Fontan J, Perrot A, et al. Isatuximab plus pomalidomide and dexamethasone in patients with relapsed/refractory multiple myeloma in real-world: the retrospective IMAGE study. Eur J Haematol. 2024;113(3):290-297.
Sanofi does not recommend the use of its products in any manner inconsistent with that described in the label available in your country. Please refer to your local product labelling information before prescribing.
Name and Presentation: SARCLISA 20 mg/mL concentrate for solution for infusion. Each vial contains 100 mg of isatuximab in 5 mL of concentrate (100 mg/5 mL) or 500 mg of isatuximab in 25 mL of concentrate (500 mg/25 mL). Isatuximab is an immunoglobulin G1 (IgG1) monoclonal antibody (mAb).
Therapeutic indications: In combination with pomalidomide and dexamethasone, for the treatment of adult patients with relapsed and refractory multiple myeloma (MM) who have received at least two prior therapies including lenalidomide and a proteasome inhibitor (PI) and have demonstrated disease progression on the last therapy. In combination with carfilzomib and dexamethasone, for the treatment of adult patients with multiple myeloma who have received at least one prior therapy. In combination with bortezomib, lenalidomide, and dexamethasone, for the treatment of adult patients with newly diagnosed multiple myeloma who are ineligible for autologous stem cell transplant.
Pediatric population: Outside its authorised indications, SARCLISA has been studied in children aged 28 days to less than 18 years of age with relapsed or refractory acute lymphoblastic or myeloid leukaemia but efficacy has not been established.
Dosage and administration: SARCLISA should be administered by a healthcare professional, in an environment where resuscitation facilities are available. Premedication should be used 15-60 minutes prior to SARCLISA infusion with the following medicinal products to reduce the risk and severity of infusion reactions: Dexamethasone 40 mg (when administered in combination with isatuximab and pomalidomide) or 20 mg (when administered in combination with isatuximab and carfilzomib; or when administered in combination with isatuximab, bortezomib, and lenalidomide) oral or intravenous, 20 mg for patients ≥75 years of age, Acetaminophen, Diphenhydramine, H2 antagonists. The recommended dose of SARCLISA is 10 mg/kg body weight administered as an intravenous infusion in combination with pomalidomide and dexamethasone or in combination with carfilzomib and dexamethasone or in combination with bortezomib, lenalidomide, and dexamethasone (isatuximab regimen). Dosing schedule in combination with pomalidomide and dexamethasone or in combination with carfilzomib and dexamethasone: cycle 1: days 1, 8, 15 and 22 (weekly), cycle 2 and beyond: days 1, 15 (every 2 weeks). Each treatment cycle consists of a 28-day period. Dosing schedule in combination with bortezomib, lenalidomide, and dexamethasone: cycle 1: days 1, 8, 15, 22 and 29, cycles 2 to 4: days 1, 15 and 29 (every 2 weeks), cycles 5 to 17: days 1 and 15 (every 2 weeks), cycles 18 and beyond: day 1 (every 4 weeks). Each treatment cycle consists of a 42-day period from cycle 1 to 4, and of a 28-day period from cycle 5. Treatment is repeated until disease progression or unacceptable toxicity.
Method of administration: SARCLISA is for intravenous use. For details on preparation and infusion rate see full SmPC.
Contraindications: Hypersensitivity to the active substance or to any of the excipients. See full SmPC for full list of excipients.
Warnings and precautions: Infusion reactions, mostly mild or moderate, were observed in 38.2% of patients treated with SARCLISA in ICARIA, and in 45.8% in IKEMA but resolved on the same day in 98% of infusions, and in 24.0% of patients treated with Isa-VRd in IMROZ and resolved the same day in 97.3% of patients. The most common symptoms of an IR included dyspnoea and chills. The most common severe sign and symptom was hypertension. Vital signs should be frequently monitored during the entire infusion and when required infusion should be interrupted or permanently discontinued in case symptoms that do not improve to grade ≤1 after infusion interruption. Serious infusion reactions including severe anaphylactic reactions have also been observed after SARCLISA administration. Most of the grade 3-4 neutropenia was reported as laboratory abnormalities. In patients treated with Isa-VRd, neutropenia was reported as a laboratory abnormality in 87.5% of patients and as an adverse reaction in 30% of patients. Neutropenic complications have been observed in 1/3 of patients treated with SARCLISA. A higher incidence of infections including grade ≥3 infections occurred with SARCLISA. Antibacterial and antiviral prophylaxis (such as herpes zoster prophylaxis) according to treatment guidelines should be considered during treatment. Patients receiving SARCLISA should be closely monitored for signs of infection. Physicians should carefully evaluate patients before and during treatment as per International Myeloma Working Group (IMWG) guidelines for occurrence of secondary primary malignancies (SPM) and treatment should be initiated as indicated. Patients should be monitored closely, and appropriate precautions taken for tumor lysis syndrome. Isatuximab binds to CD38 on red blood cells (RBCs) and may result in a false positive indirect antiglobulin test (indirect Coombs test). This interference with the indirect Coombs test may persist for at least 6 months after the last infusion of SARCLISA. Patient should have blood type and screen tests performed prior to the first infusion of Isatuximab and should be monitored for theoretical risk of haemolysis. For details in tests interference see full SmPC.
Drug interactions: Isatuximab has no impact on the pharmacokinetics of pomalidomide or carfilzomib, or bortezomib, or lenalidomide and vice versa. Isatuximab may interfere with serological testing and with Serum Protein Electrophoresis and Immunofixation assays. In patients with persistent very good partial response, where isatuximab interference is suspected, consider using a validated isatuximab-specific IFE assay to distinguish isatuximab from any remaining endogenous M protein in the patient’s, to facilitate determination of complete response.
Fertility, pregnancy and lactation: Women of childbearing potential treated with isatuximab should use effective contraception during treatment and for 5 months after cessation of treatment. The use of isatuximab in pregnant women is not recommended since there are no available data.
Undesirable effects: Observed in patients treated with isatuximab in combination with pomalidomide and dexamethasone: Infections/infestations: very common: pneumonia, upper respiratory tract infection, bronchitis: common: Herpes zoster. Neoplasms benign, malignant and unspecified: common: skin cancer, solid tumour (non-skin cancer): uncommon: haematology malignancy. Blood/lymphatic system disorders: very common: neutropenia, thrombocytopenia, common: febrile neutropenia, anaemia, unknown frequency: lymphopenia. Metabolism and nutrition disorders: very common: decreased appetite. Cardiac disorders: common: atrial fibrillation. Respiratory, thoracic and mediastinal disorders: very common: dyspnoea. Gastrointestinal disorders: very common: diarrhoea, nausea, vomiting. Investigations: common: weight decreased. Injury, poisoning and procedural complications: very common: infusion reaction. Immune system disorders: uncommon: anaphylactic reaction. Observed in patients treated with isatuximab in combination with carfilzomib and dexamethasone: Infections/infestations: very common: pneumonia, upper respiratory tract infection, bronchitis: common: Herpes Zoster. Vascular disorder: very common: hypertension. Neoplasms benign, malignant and unspecified: common: Skin cancers and solid tumors non-skin cancers. Blood/lymphatic system disorders: common: neutropenia, anaemia, thrombocytopenia, unknown frequency: lymphopenia. Respiratory, thoracic and mediastinal disorders: very common: dyspnoea and cough. Gastrointestinal disorders: very common: diarrhoea and vomiting. General disorders and administration site conditions: very common: Fatigue. Injury, poisoning and procedural complications: very common: infusion reaction. Immune system disorders: uncommon: anaphylactic reaction. Reported in patients with multiple myeloma treated with isatuximab in combination with bortezomib, lenalidomide, and dexamethasone: Infections/infestations: very common: pneumonia, bronchitis, Covid-19. Neoplasms benign, malignant and unspecified: common: skin cancer, solid tumour, uncommon: haematology malignancy. Blood and lymphatic system disorders: very common: neutropenia, thrombocytopenia, common: anaemia, not known: lymphopenia. Immune system disorders: uncommon: anaphylactic reaction. Eye disorders: very common: cataract. Gastrointestinal disorders: very common: diarrhoea, common: vomiting. General disorders and administration site conditions: very common: fatigue. Injury, poisoning and procedural complications: very common: infusion reaction.
Pharmacotherapeutic group: Antineoplastic agents, monoclonal antibodies, ATC code: L01FC02.
List of excipients: Sucrose, Histidine hydrochloride monohydrate, Histidine, Polysorbate 80 and Water for injections.
Legal classification: Prescription Only Medicine.
Marketing authorization holder: Sanofi Winthrop Industrie, 82, avenue Raspail, 94250 Gentilly, France.
Date of last revised: March 2025.
Abbreviated Prescribing Information based on the EU SmPC as of February 2025.
Before prescribing always refer to your full local prescribing information as this information may vary from country to country
MAT-GLB-2101917-v10.0-03/2025
