Thymoglobulin May Suppress the Immune Response through Multiple Pathways1
The mechanism of action by which Thymoglobulin suppresses the immune response is not fully understood, but may include1:
- T-cell clearance from the circulation
- Modulation of T-cell activation, homing, and cytotoxic activities
See How it Works
View a full depiction of the mechanisms of acute rejection and how Thymoglobulin may help with prevention or treatment.
Click here for full Prescribing Information.
Thymoglobulin includes antibodies against T-cell markers such as CD2, CD3, CD4, CD8, CD11a, CD18, CD25, CD44, CD45, HLA-DR, HLA Class I heavy chains, and β2 micro-globulin.1
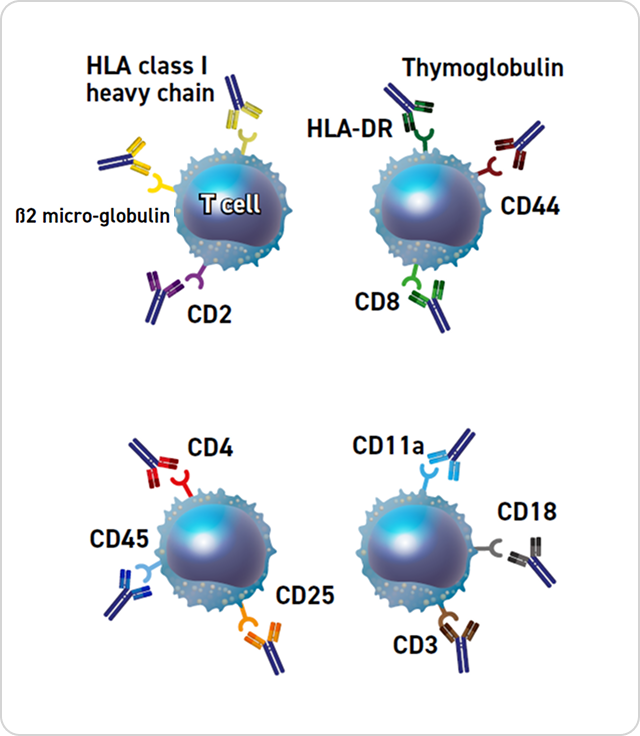
Potential Mechanisms of Action of Thymoglobulin
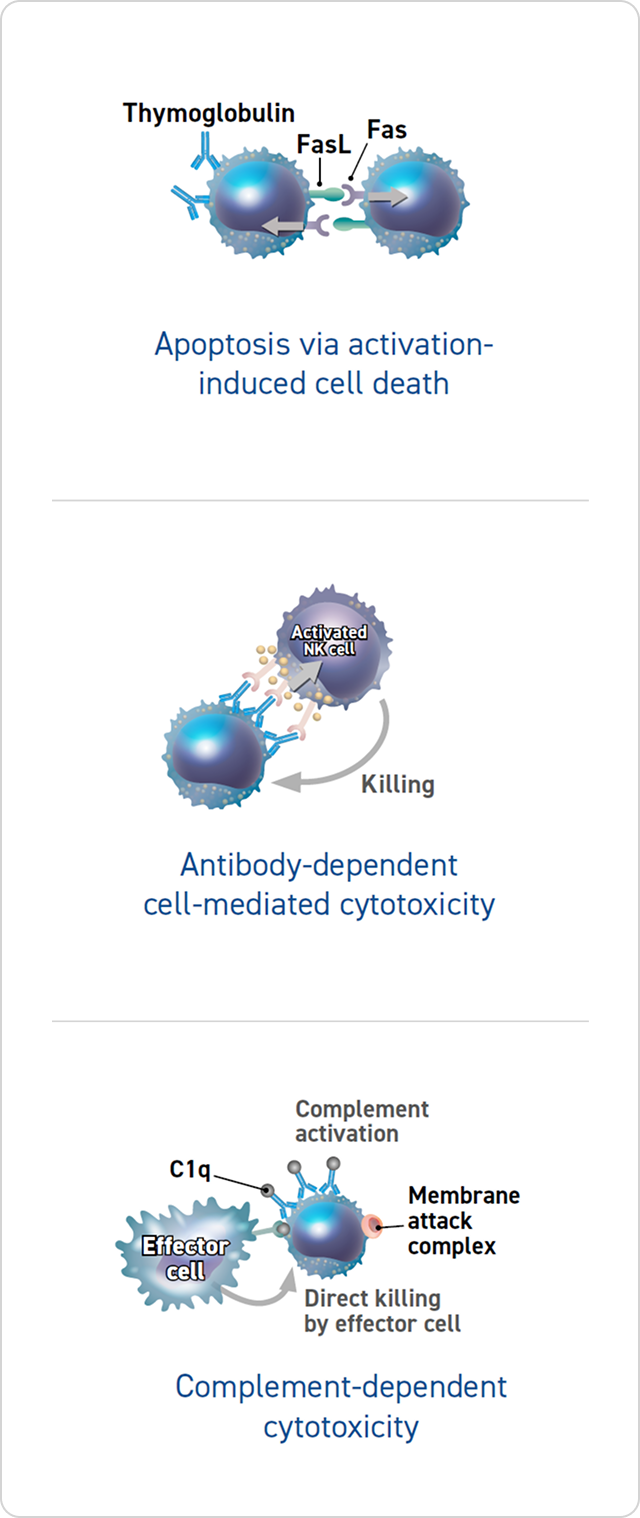
T-Cell Clearance From the Circulation
Thymoglobulin may deplete T cells through 3 different mechanisms:
- Apoptosis via activation-induced cell death2-4
- Antibody-dependent cell-mediated cytotoxicity4
- Complement-dependent cytotoxicity4
The clinical significance of these mechanisms is unknown.
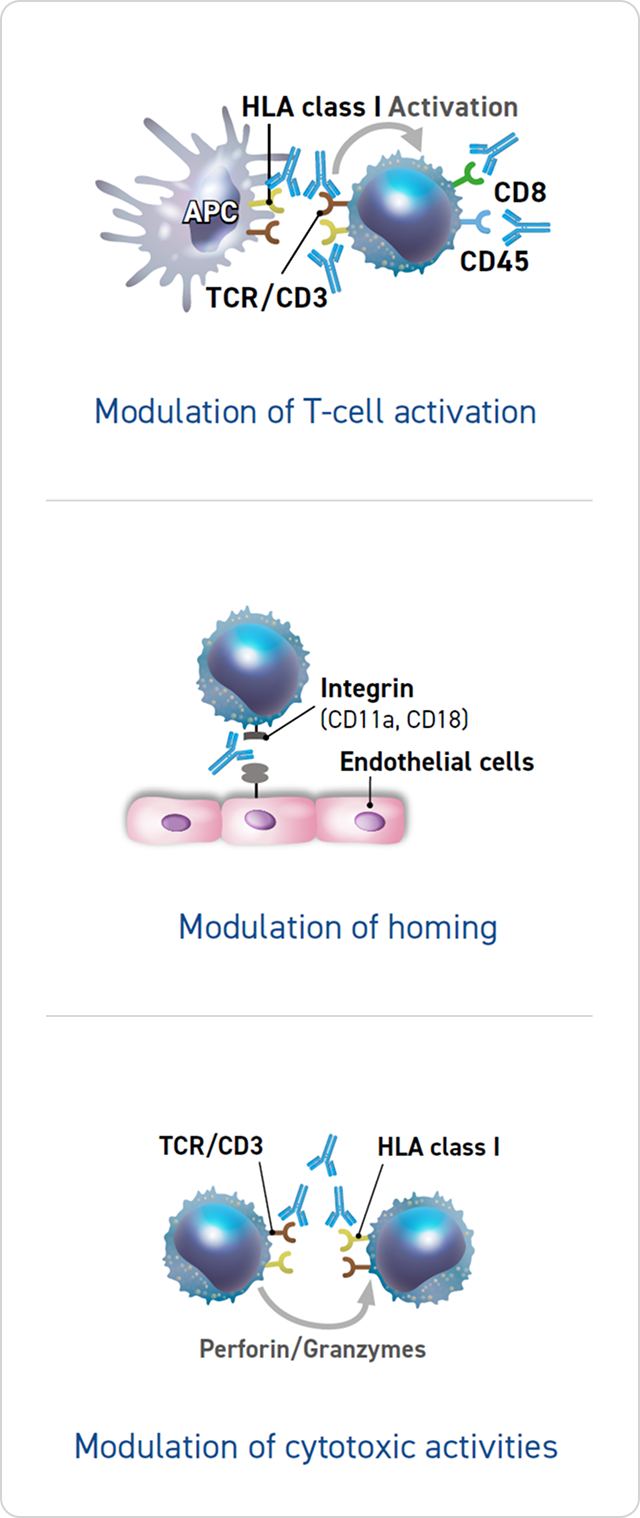
Modulation of T-Cell Activation, Homing, and Cytotoxic Activities
Thymoglobulin may modulate immunosuppression through the following mechanisms:
- Downmodulation of the expression of several molecules that control T-cell activation (ie, TCR/CD3, CD4, CD8, CD25, CD45,
HLA class I, HLA-DR)1,5 - Binding to selectins and integrin families (CD11a, CD18), thereby preventing T-cell homing5
- Prevention of cytotoxic activities1,5,6
The clinical significance of these mechanisms is unknown.
Thymoglobulin Is the Only Polyclonal T-Cell–Depleting Agent That Is FDA Approved for Prevention of Acute Rejection1,7
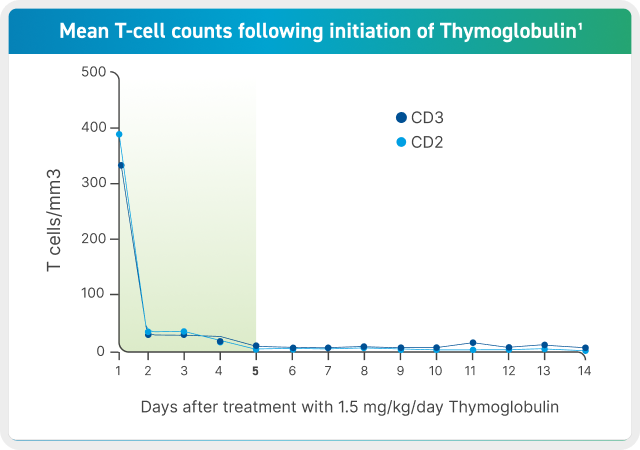
- Based on data collected from 12 patients in the phase 3 trial, T-cell depletion is usually observed within a day from initiating Thymoglobulin therapy.1
Important Safety Information
Abbreviations: CD, cluster of differentiation; FDA, Food and Drug Administration; FasL, Fas ligand; HLA, human leukocyte antigen; HLA-DR, human leukocyte antigen-D related; NK, natural killer cells; TCR, T-cell receptor.
References:
1. Thymoglobulin [prescribing information]. Cambridge, MA: Genzyme Corporation; 2026.
2. Woodside KJ, Hu M, Gugliuzza KK, Hunter GC, Daller JA. T-lymphocyte apoptosis is increased by non-interleukin-2-dependent induction in human mixed lymphocyte cultures. Transplantation Proc. 2005;37(4):1949-1952.
3. Bonnefoy-Berard N, Genestier L, Preville X, Revillard JP. TNFα and CD95-L contribute to apoptosis of activated lymphocytes triggered by ATGs. Transplantation Proc. 1999;31(1-2):775-777.
4. Genestier L, Fournel S, Flacher M, Assossou O, Revillard JP, Bonnefoy-Berard N. Induction of Fas (Apo-1, CD95)-mediated apoptosis of activated lymphocytes by polyclonal antithymocyte globulins. Blood. 1998;91(7):2360-2368.
5. Mueller TF. Mechanisms of action of Thymoglobulin. Transplantation. 2007;84(11S):S5-S10.
6. Krepsova E, Tycova I, Sekerkova A, et al. Effect of induction therapy on the expression of molecular markers associated with rejection and tolerance. BMC Nephrol. 2015;16:146.
7. Atgam [prescribing information]. New York, NY: Pfizer Inc; 2023.
![Graphic with the text 'Thymoglobulin® [Anti‑thymocyte Globulin (Rabbit)'] with antibody symbol icon on a blue background with kidney line patterns behind a man and a small disclaimer 'Not an actual patient'.](https://pro.campus.sanofi/dam/jcr:b1604342-1be0-472c-bc4b-6027c9677be8/SAN-THYMO%20-%20mobile%20-%20main%20banner.png)