Both Recipient and Donor Risk Factors should be Considered when Evaluating Risk of Acute Rejection1,2
Many Factors Are Associated With an Increased Risk of Acute Rejection1-7
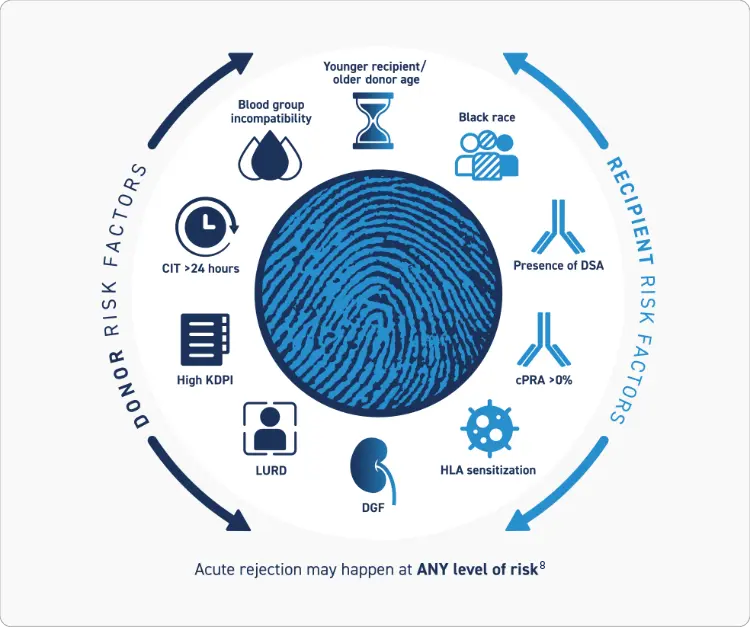
Patients with 1 or more risk factors may be considered to be at high risk of acute rejection1
Protection against Acute Rejection is a Critical Step to Preserve Kidney Function9
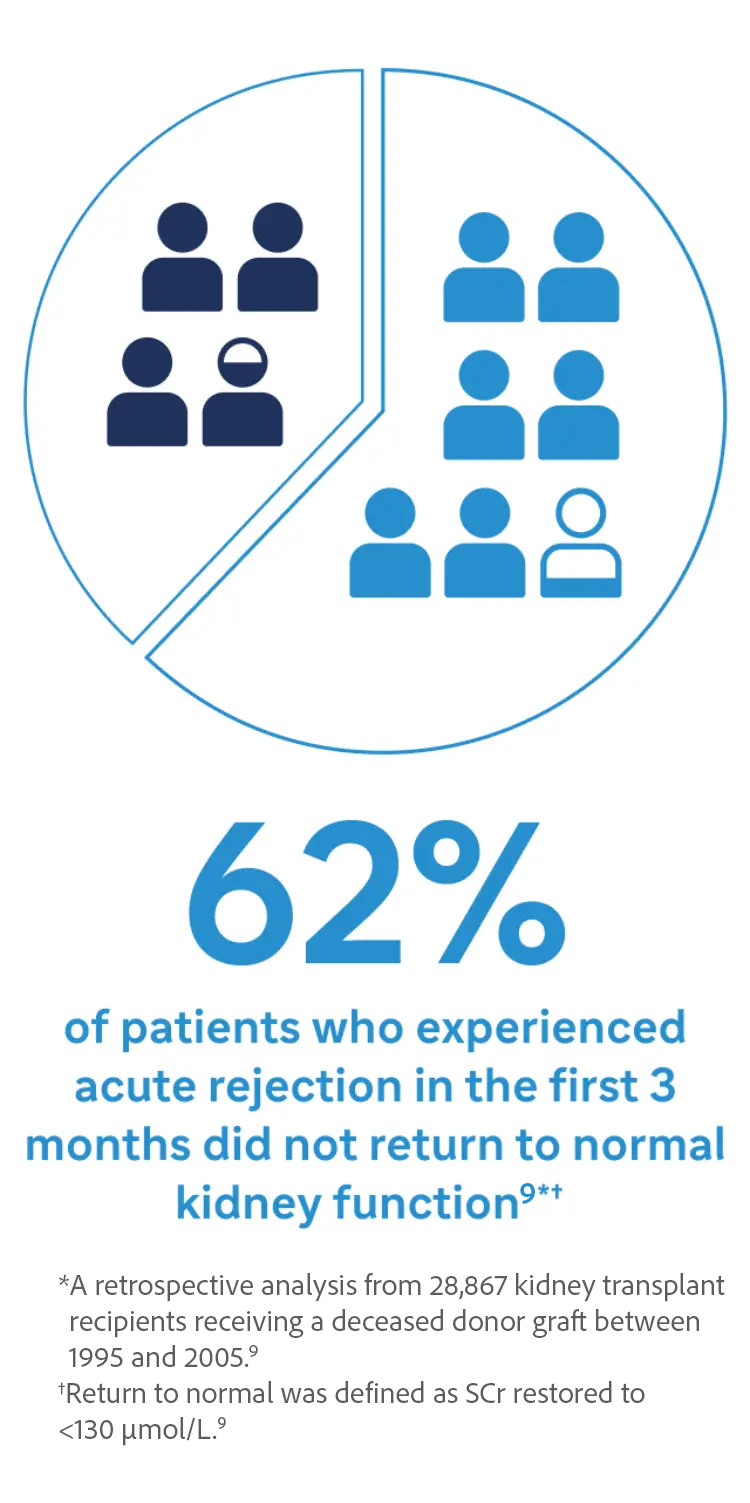
Potential Consequences of Acute Rejection to the Patient and Transplant Center
Patient
- Reduced quality of life and increased emotional burden10,11
- Return to long-term dialysis10,12,13
- Retransplantation10
- Increased costs10
Transplant Center
- Increased likelihood of regulatory scrutiny14
- Increased costs/resources associated with13,15
- Treatment of acute rejection
- Additional biopsies
- Hospitalization (return to dialysis)
- Graft failure
What impact does acute rejection have at your center?
PREVIOUS
← Overview
Important Safety Information
Abbreviations: CIT, cold ischemia time; cPRA, calculated panel reactive antibodies; DGF, delayed graft function; DSA, donor-specific antibodies; HLA, human leukocyte antigen; KDPI, Kidney Donor Profile Index; LURD, living unrelated donor; SCr, serum creatinine.
References:
1. Lebranchu Y, Baan C, Biancone L, et al. Pretransplant identification of acute rejection risk following kidney transplantation. Transplant Int. 2013;27(2):129-138.
2. Kidney Disease: Improving Global Outcomes (KDIGO) Transplant Working Group. KDIGO clinical practice guideline for the care of kidney transplant recipients. Am J Transplantation. 2009;9(suppl 3):S1-S155.
3. Treat E, Chow EKH, Peipert JD, et al. Shipping living donor kidneys and transplant recipient outcomes. Am J Transplantation. 2018;18(3):632-641.
4. Øien CM, Reisæter AV, Leivestad T, Dekker FW, Line PD, Os I.Living donor kidney transplantation: the effects of donor age and gender on short- and long-term outcomes. Transplantation. 2007;83(5):600-606.
5. Jackson KR, Holscher C, Motter JD, et al. Posttransplant outcomes for cPRA-100% recipients under the new kidney allocation system. Transplantation. 2020;104(7):1456-1461.
6. Ravindra KV, Sanoff S, Vikraman D, et al. Lymphocyte depletion and risk of acute rejection in renal transplant recipients at increased risk for delayed graft function. Am J Transplantation. 2019;19(3):781-789.
7. Fuller TF, Feng S, Brennan TV, Tomlanovich S, Bostrom A, Freise CE. Increased rejection in living unrelated versus living related kidney transplants does not affect short term function and survival. Transplantation. 2004;78(7):1030-1035.
8. National Kidney Foundation. Kidney transplant: what you need to know. Accessed January 7, 2026. https://www.kidney.org/sites/default/files/kidneytransplant_updated.pdf
9. Opelz G, Döhler B; Collaborative Transplant Study Report. Influence of time of rejection on long-term graft survival in renal transplantation. Transplantation. 2008;85(5):661-666.
10. Ndemera H, Bhengu B. Factors contributing to kidney allograft loss and associated consequences among post kidney transplantation patients. Health Sci J. 2017;11(3):504.
11. Ouellette A, Achille MA, Vachon M. Psychological impact of kidney graft failure and implications for the psychological evaluation of retransplant candidates. Dial Transplantation. 2006;35(6):354-361.
12. Koo EH, Jang HR, Lee JE, et al. The impact of early and late acute rejection on graft survival in renal transplantation. Kidney Res Clin Pract. 2015;34(3):160-164.
13. Schnitzler MA, Johnston K, Axelrod D, Gheorghian A, Lentine KL. Associations of renal function at 1-year after kidney transplantation with subsequent return to dialysis, mortality, and healthcare costs. Transplantation. 2011;91(12):1347-1356.
14. Snyder JJ, Salkowski N, Wey A, et al. Effects of high-risk kidneys on scientific registry of transplant recipients program quality reports. Am J Transplantation. 2016;16(9):2646-2653
15. Łabus A, Mucha K, Kulesza A, Fliszkiewicz M, Paczek L, Niemczyk M. Costs of treatment of acute antibody-mediated rejection in kidney transplant recipients. Transplantation Proc. 2022;54(4):968-971.
