Dosing and administration instructions
Administration
TZIELD is administered by intravenous infusion over a minimum of 30 minutes, using body surface area-based (BSA) dosing1
Dosage form and strength
2 mg/2 mL (1 mg/mL), clear and colourless solution in a single-dose vial1
No same-day dosing
Do NOT administer two doses on the same day1
If an infusion is missed
Resume by administering all remaining doses on consecutive days to complete the 14-day course1
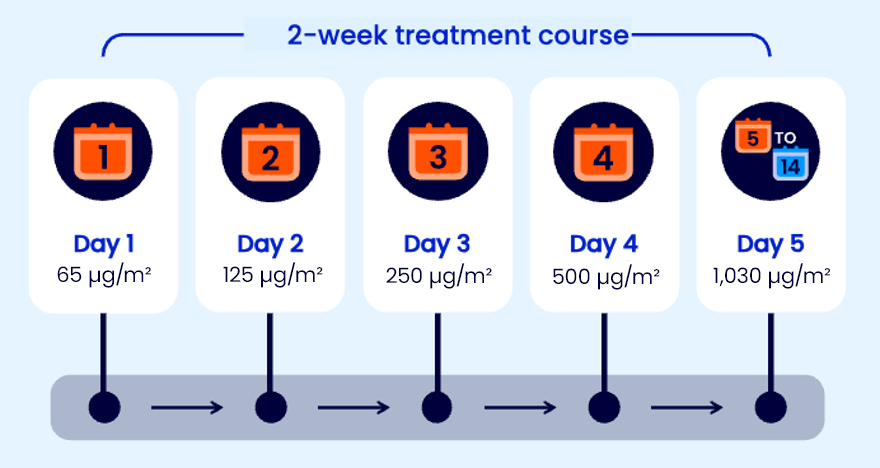
Recommended dosing schedule for TZIELD
The recommended TZIELD dosage for adults and paediatric patients aged 8 years and older uses body surface area (BSA)-based dosing and is administered according to the following regimen:1
Calculating your patients' BSA using the Mosteller formula2

- When calculating BSA, round to the 100th using standard rounding rules (example: 0.93 m2)
- Based on BSA dosing requirements, two vials maybe needed for some individuals (BSA >1.94 m2) for days 5–141
Use this interactive tool below to calculate individual dosing and support safe and confident infusion delivery in your clinical practice.
TZIELD is a concentrate solution for infusion. Each vial contains 2 mg of teplizumab in 2 mL of concentrate. The product must be diluted prior to use.1
TZIELD should be administered by a healthcare professional with access to appropriate medical support to manage potential severe adverse reactions.
Patient selection
Select adult and paediatric patients 8 years of age and older for TZIELD treatment who have a diagnosis of Stage 2 T1D1
- Confirm Stage 2 T1D by documenting:
- At least two positive pancreatic islet cell autoantibodies (AAbs)
- Dysglycaemia without overt hyperglycaemia
- Ensure the clinical history of the patient does not suggest Type 2 diabetes (T2D)
Laboratory evaluation and vaccinations prior to treatment
Prior to initiation of TZIELD, there are a number of laboratory evaluations required to ensure that TZIELD is suitable for patients. All age-appropriate vaccinations must also be administered before treatment.1
Prior to initiating TZIELD, obtain a complete blood count and liver enzyme tests
Use of TZIELD is not recommended in patients with:
- Lymphocyte count <109 lymphocytes/L
- Haemoglobin <100 g/L
- Platelet count <150 x 109 platelets/L
- Absolute neutrophil count <1.0 x 109 neutrophils/L in those of African descent and <1.5 x 109 neutrophils/L in all other groups
- Elevated alanine aminotransferase (ALT) or aspartate aminotransferase (AST) >2 x upper limit of normal (ULN) or bilirubin greater than 1.5 times ULN
- Laboratory or clinical evidence of acute infection with Epstein-Barr virus (EBV) or cytomegalovirus (CMV)
- Active serious infection or chronic active infection other than localised skin infections
Administer all age-appropriate vaccinations prior to starting TZIELD:
- Administer live-attenuated (live) vaccines at least 8 weeks prior to treatment
- Administer inactivated (killed) vaccines or messenger ribonucleic acid (mRNA) vaccines at least 2 weeks prior to treatment
Get in Touch with Us
Questions? Leave your details and we'll reach out to you at your preferred time.
Get in touchINDICATION: TZIELD is indicated to delay the onset of Stage 3 T1D in adult and paediatric patients 8 years of age and older with Stage 2 T1D.1
*The dosing schedule in TN-10 was different to the recommended dosing schedule in the SmPC.1
AAbs, autoantibodies; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BSA, body surface area; CMV, cytomegalovirus; EBV, Epstein-Barr virus; mRNA, messenger ribonucleic acid; NICE, National Institute for Health and Care Excellence; NSAID, non-steroidal anti-inflammatory drug; SmPC, Summary of Product Characteristics; T1D, Type 1 diabetes; T2D, Type 2 diabetes; UK, United Kingdom.
- TZIELD® (teplizumab) UK Summary of Product Characteristics. 2025.
- Mosteller RD. N Engl J Med. 1987; 317(17): 1098.
MAT-XU-2500768 (v1.0) March 2026
