- Article
- Source: Campus Sanofi
- Sep 17, 2025
Burden of AT1D
PERCENTAGE OF PSYCHIATRIC / MENTAL HEALTH CONDITIONS1
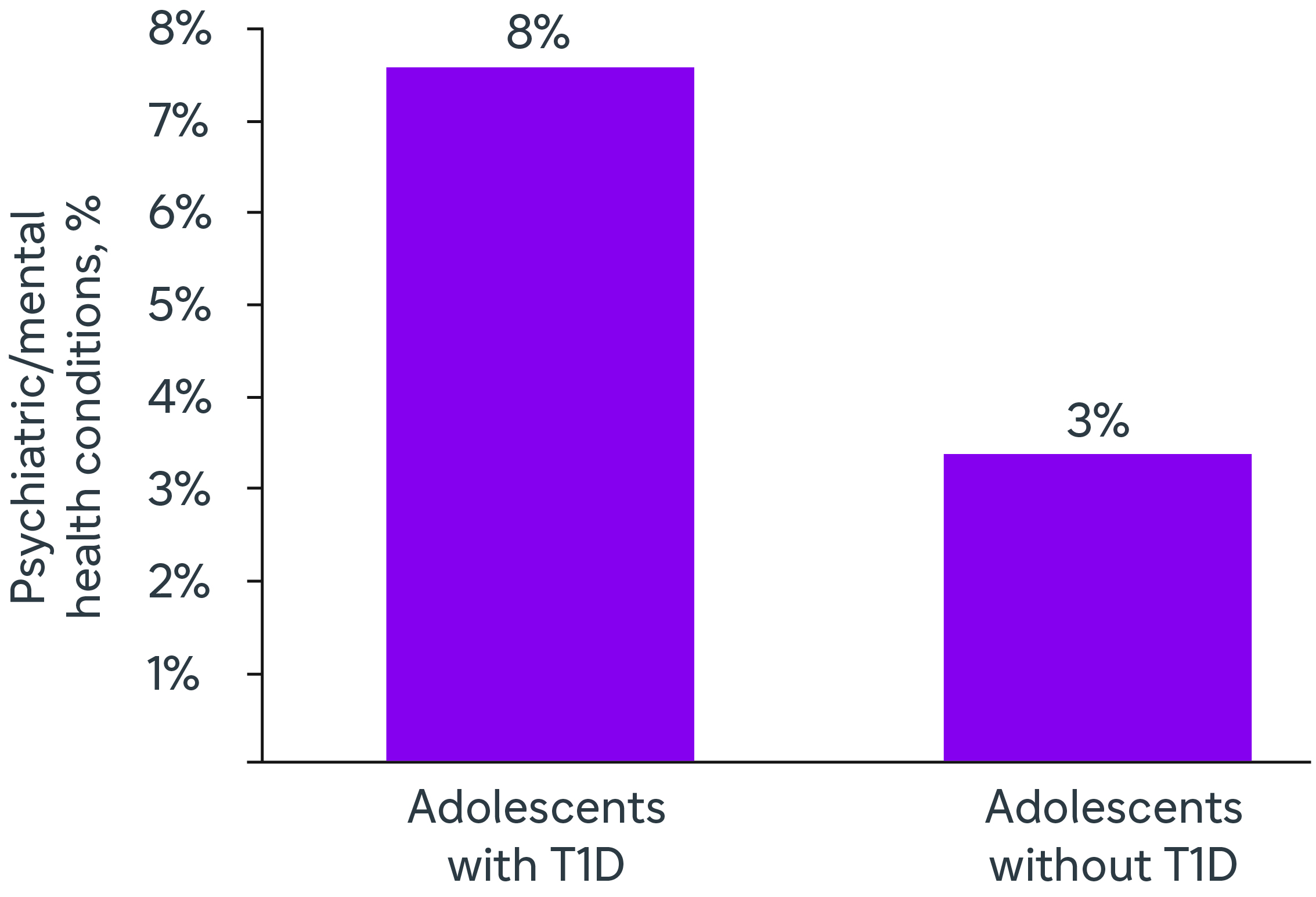
QoL Impacts
- The ADA and EASD recommend regular screening for psychosocial issues and diabetes-related distress beginning in childhood2,6
- Once stage 3 T1D develops, patients and their caregivers face a substantial humanistic burden associated with the condition and its management. Across age groups, individuals with T1D have reduced quality of life (QoL) compared with the general population3,4,5
Onset of stage 3 aT1D at a younger age is associated with increased CVD risk and early mortality.4
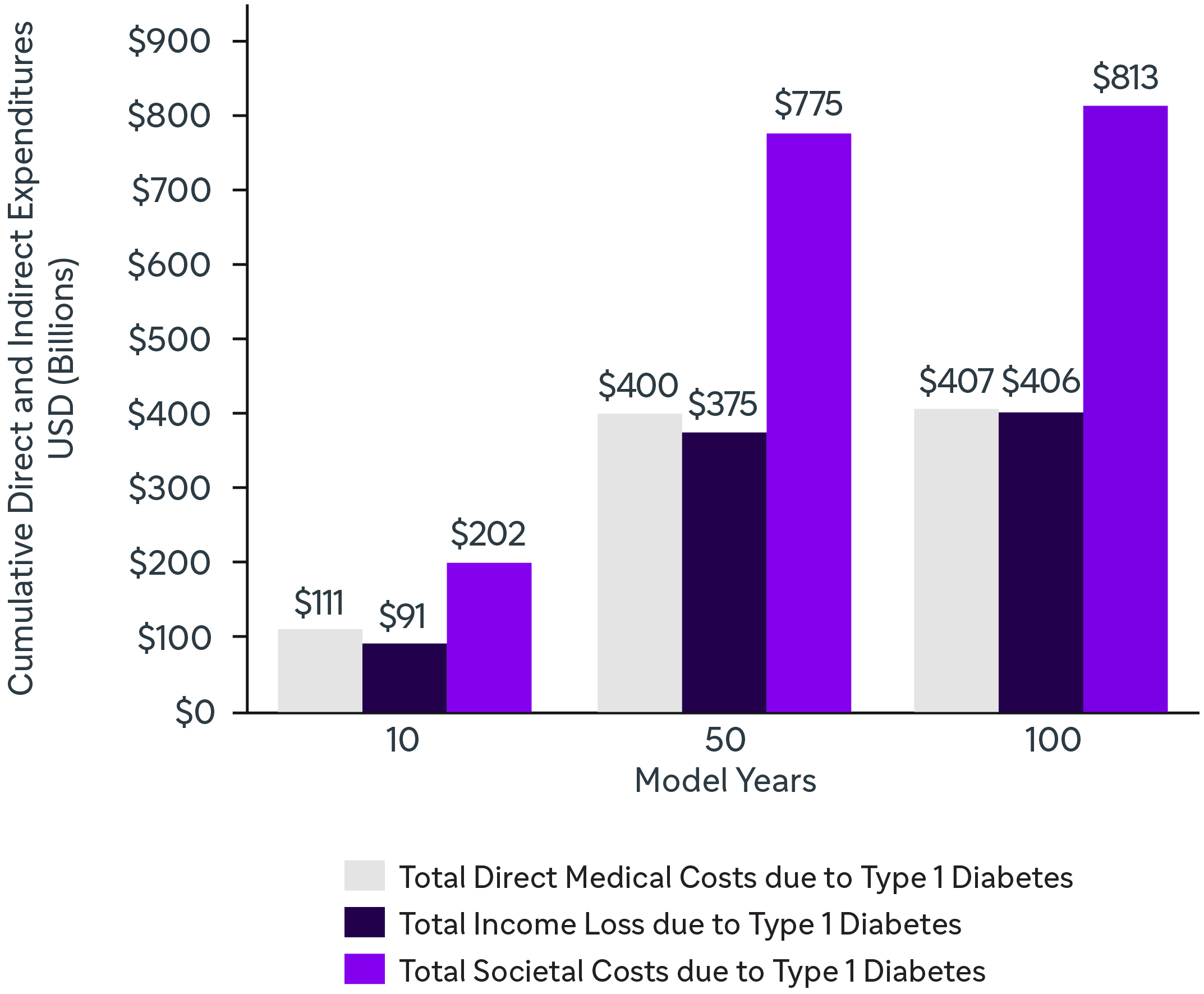
Lifelong management of aT1D drives significant increases in both direct (HCRU) and indirect costs (productivity loss, caregiver burden).5
PREDICTED LIFETIME CUMULATIVE COSTS OF TID IN THE US (USD, 2016)*
CUMLATIVE COSTS OF TID IN FR (LOCAL CURRENCY, 2023)
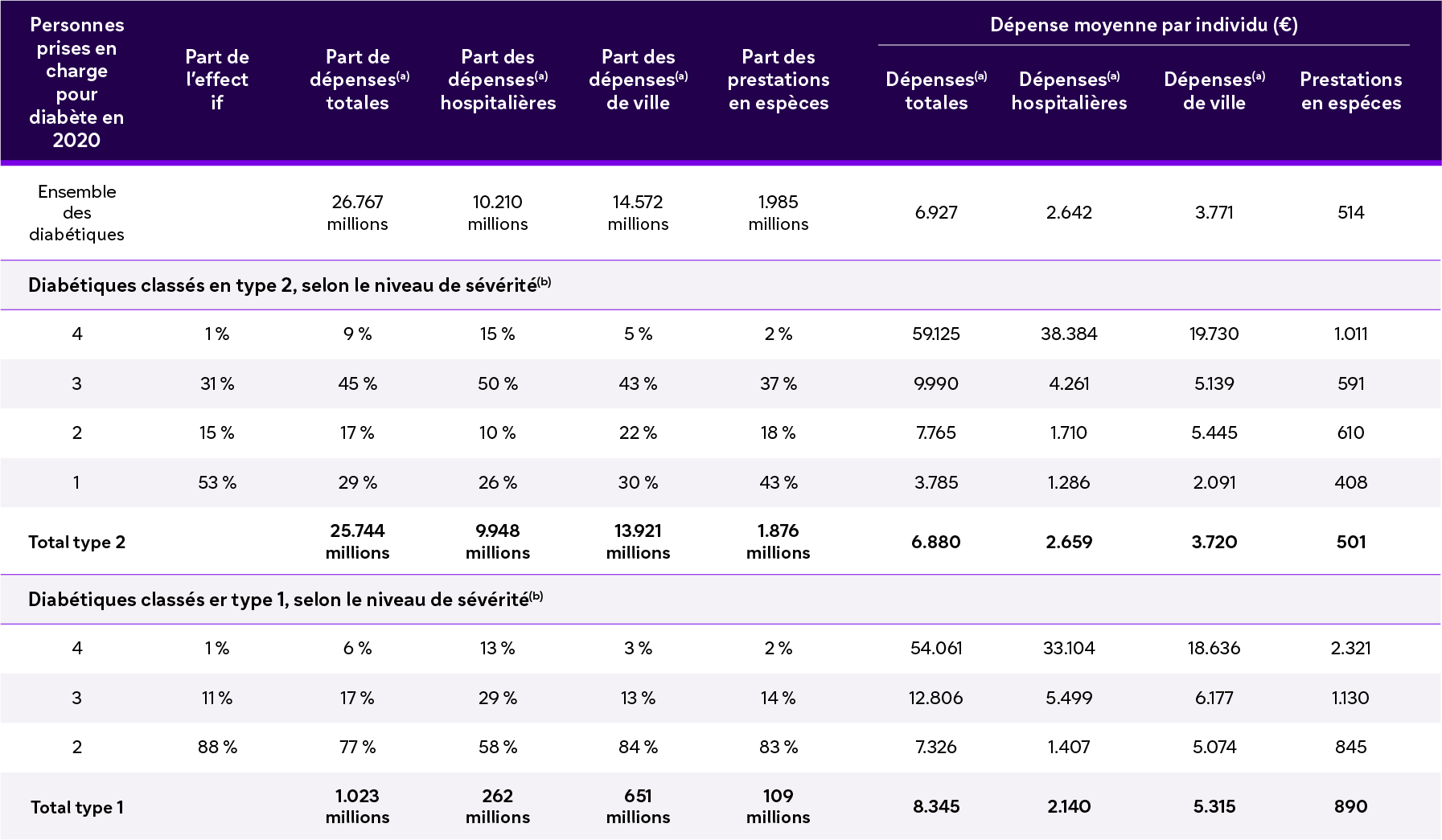
(a) Il s'agit de l'ensemble des dépenses et pas spécifiquement de celles liées à la prise en charge du diabète
(b) Niveaux croissants de sévérité, en fonction des complications liées au diabète; la définition de chaque niveau est détaillée dans l'encadré méthodologique
- Liu S, Ludvigsson JF, Lichtenstein P, et al. Educational Outcomes in Children and Adolescents With Type 1 Diabetes and Psychiatric Disorders. JAMA Netw Open. Apr 3 2023;6(4):e238135. doi:10.1001/jamanetworkopen.2023.8135
- Besser REJ, Bell KJ, Couper JJ, et al. ISPAD Clinical Practice Consensus Guidelines 2022: Stages of type 1 diabetes in children and adolescents. Pediatr Diabetes. Dec 2022;23(8):1175-1187. doi:10.1111/pedi.13410
- Sparring V, Nyström L, Wahlström R, Jonsson PM, Ostman J, Burström K. Diabetes duration and health-related quality of life in individuals with onset of diabetes in the age group 15-34 years - a Swedish population-based study using EQ-5D. BMC Public Health. Apr 22 2013;13:377. doi:10.1186/1471-2458-13-377
- Bronner MB, Peeters MAC, Sattoe JNT, van Staa A. The impact of type 1 diabetes on young adults' health-related quality of life. Health Qual Life Outcomes. May 12 2020;18(1):137. doi:10.1186/s12955-020-01370-8
- Rechenberg K, Koerner Ms R. General Anxiety, Diabetes-Specific Anxiety, and Quality of Life in Adolescents With Type 1 Diabetes. Sci Diabetes Self Manag Care. Dec 2022;48(6):469-475. doi:10.1177/26350106221125689
- Holt RIG, DeVries JH, Hess-Fischl A, et al. The Management of Type 1 Diabetes in Adults. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. Nov 2021;44(11):2589-2625. doi:10.2337/dci21-0043
GULF:
One JLT Building, Level 3, Jumeirah Lake Towers, PO Box 53899, Dubai, UAE .
For further medical information, please contact: For UAE ✆ 800 MEDICAL Toll-Free Number.
For all Gulf countries ✆ +971 45 50 38 63 or email: medical-information.gulf@sanofi.com.
Full prescribing information is available upon request.
To Report adverse events please call: +971 561747001 or email Gulf.Pharmacovigilance@sanofi.com
www.sanofi.com
KSA:
To report any side effect(s):
Saudi Arabia: The National Pharmacovigilance and Drug Safety Centre (NPC
SFDA call center: 19999 • E-mail: npc.drug@sfda.gov.sa • Website: https://ade.sfda.gov.sa/
Full Prescribing Information is available upon request: SANOFI, Kingdom of Saudi Arabia, P.O. Box 9874, Jeddah 21423, K.S.A.
Tel: +966-12-669-3318, Fax: +966-12-663-6191
For Medical Information, please contact: +966-12-669-3318, ksa.medicalinformation@sanofi.com
For Pharmacovigilance, please contact: +966-54-428-4797, ksa_pharmacovigilance@sanofi.com
To report any Product Technical Complaint, please contact SANOFI Quality Department:
Email: quality.greatergulf@Sanofi.com , www.sanofi.com.sa
MAT-KW-2600001-V1-Feb 2026