- Article
- Source: Campus Sanofi
- Sep 17, 2025
AT1D Early Detection
Early detection programs reduce rates of DKA by up to 90%, preventing severe complications and reducing hospitalization at Stage 3 diagnosis as shown in multiple studies
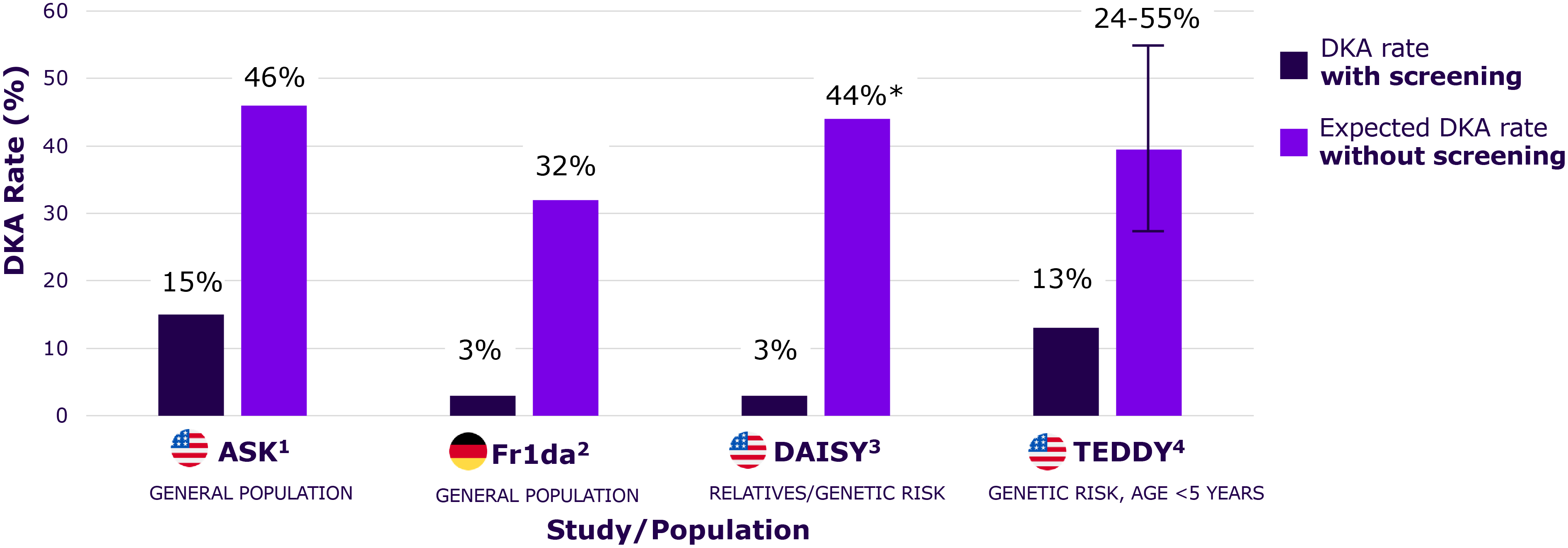
Significant short-term morbidity and cost, as well as long-term impacts on neurofunction and glycemic control
*Hospitalization rate, which was mainly driven by DKA in the control patients, was reported rather than DKA in DAISY. DKA, diabetic ketoacidosis; T1D, Type 1 diabetes.
Rates are dramatically reduced with screening and education
People that test positive can monitor, prepare and manage progression more effectively resulting in improved beta cells activity & glycemic control at symptoms onset5
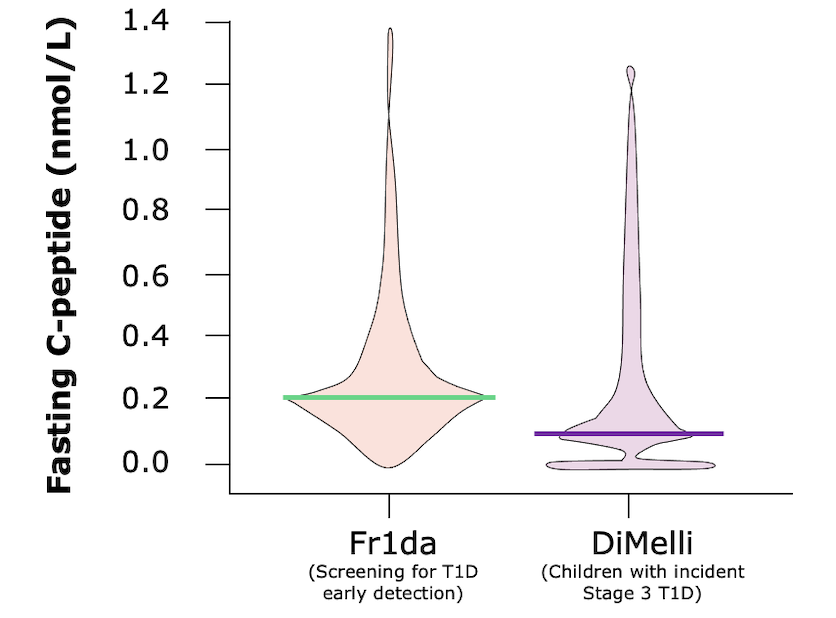
FASTING C-PEPTIDE AT CLINICAL MANIFESTATION***
HBA1C AT CLINICAL MANIFESTATION***
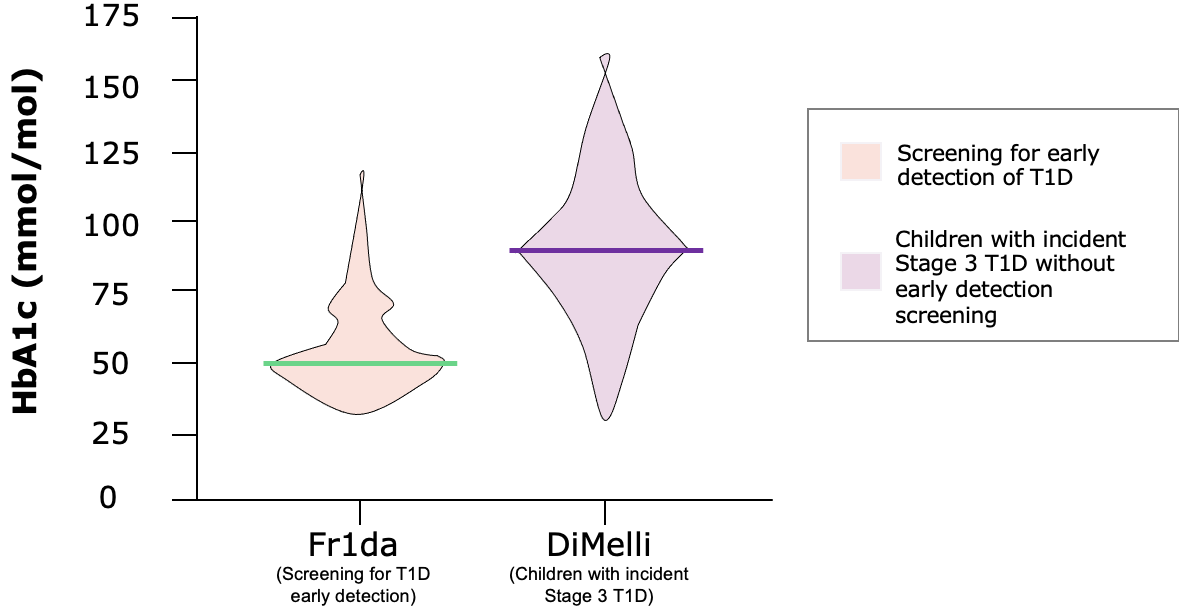
Children diagnosed with pre-clinical T1D through public health screening had milder diabetes at clinical manifestation following education and monitoring
The Fr1da study was a program screening over 165,000 children aged 2–5 years for islet autoantibodies in Bavaria, Germany as a model to evaluate population-based screening for early detection of T1D. The main purpose of the screening is to diagnose presymptomatic T1D so that affected children and their families can be educated and monitored, and so that patients can begin insulin therapy early enough to prevent serious metabolic derangements at the onset of clinical disease. The primary outcome was presymptomatic T1D, defined by 2 or more islet autoantibodies, with categorization into stages 1 (normoglycemia), 2 (dysglycemia), or 3 (clinical) T1D. Secondary outcomes were the frequency of DKA and parental psychological stress, assessed by the Patient Health Questionnaire-9 (range, 0–27; higher scores indicate worse depression; ≤4 indicates no to minimal depression; >20 indicates severe depression).1,2 The DiMelli study was a pediatric diabetes registry that enrolled children with incident Stage 3 T1D diagnosed through the same clinical centers in Bavaria as in the Fr1da study, but without prior screening for islet autoantibodies.2
Most patients will test negative which offers them & their care givers peace of mind especially for family relatives living with T1D.6
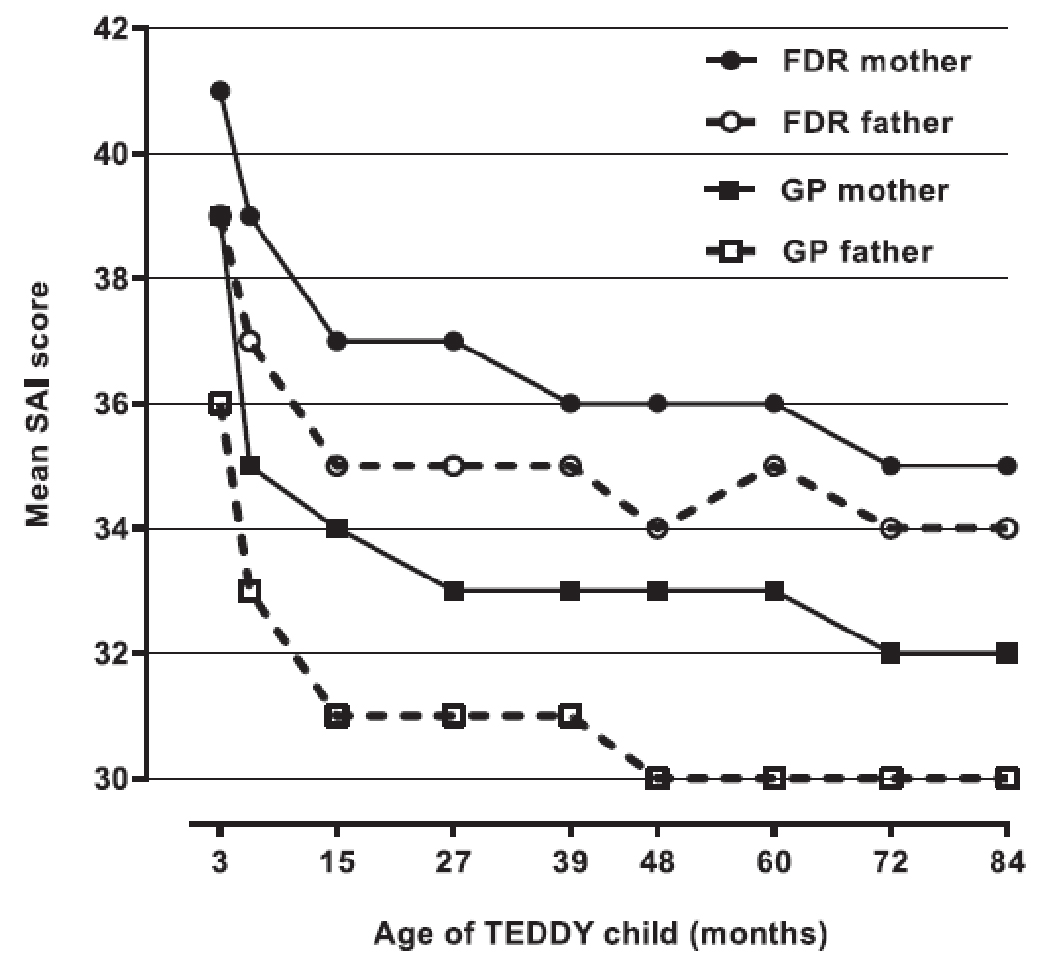
Figure 1-Parent SAl scores across time for GP and FDR children with IA - test results.
- Parents showed high levels of anxiety in response to their child's increased risk of T1D. Mothers were more anxious than fathers and parents with diabetes in their family were more anxious than parents with no history.
- In response to repeated negative test results, parent anxiety declined to normal levels.
RESULTS
At study inception, parents showed high levels of anxiety in response to their child's increased genetic type 1 diabetes risk; mothers were more anxious than fathers, and parents with diabetes in the family were more anxious than parents with no family history. In response to repeated IA-negative (IA-) test results, parent anxiety declined to normal levels. Anxiety increased in parents faced with an lA-positive (IA+) test result. Parents faced with two or more types of lA+ test results showed particularly high levels of anxiety (all P < 0.001).
Early detection can further reduce the number of T1D patients commonly misdiagnosed as having T2D7

Misdiagnosis can delay proper insulin replacement therapy, resulting in prolonged hyperglycaemia and increasing risk of DKA
Diabetes screening is associated with similar cost-effective profile as other prevention strategies
|
Cost per intervention to avoid one event | |||
| Screening of diabetes to prevent DKA | RSV drug to prevent hospitalization8 | Immunization to prevent Meningitis B9 | |
| Cost (€) |
30 (estimate) |
350* |
167,4* |
| Positive rate |
0,30% |
|
|
| Cost per individual under protection |
10 000 |
350* |
167,4* |
| Reduction of event by intervention |
80,0% |
82,0% |
79% |
| Number of event without intervention |
40,0% |
1,9% |
|
| Number of event with intervention |
10,0% |
0,3% |
|
| Cost per event avoid |
27 778 |
22 702 |
2 000 000 (Closer to rare disease approach) |
* French prices
Screening to avoid DKA alone might not be cost effective, but including lifetime benefits leads to cost effectiveness
Lifetime cost-effectiveness results10
| Screening strategy |
DKA ratio |
Incremental population average HbA1c per patient |
Other diabetes complication costs over a lifetime** |
Incremental Effectiveness (life years) |
Incremental total cost-effectiveness ratio |
| No screening |
58% |
Reference |
$284,000,000 |
Reference |
Reference |
| Ages 2 and 6 IAB only |
29.6% |
-0.24% |
$282,000,000 |
363 |
$50,000 per life year gained |
| Ages 2 and 6 IAB + GRS high risk |
33.7% |
-0.21% |
$283,000,000 |
363 |
$6,000 per life year gained |
*Both cost effective at <$100,000 per life year gained****
**All costs are in 2024 USD
***Total costs include screening costs for N=100,000 children and adolescents aged 0-15, T1D onset costs (with and without DKA), and lifetime diabetes complication-related casts for N=504 T1D cases eventually diagnosed within the first 15 years and followed for a lifetime.
****David J. Vanness, James Lomas, Hannah Ahn. A Health Opportunity Cost Threshold for Cost-Effectiveness Analysis in the United States. Ann Intern Med.2021:174:25-32.
Results are preliminary and subiect to change
People living with certain autoimmune diseases, such as celiac and thyroid, are also more likely to develop aT1D (per ADA, BreakthroughT1D).
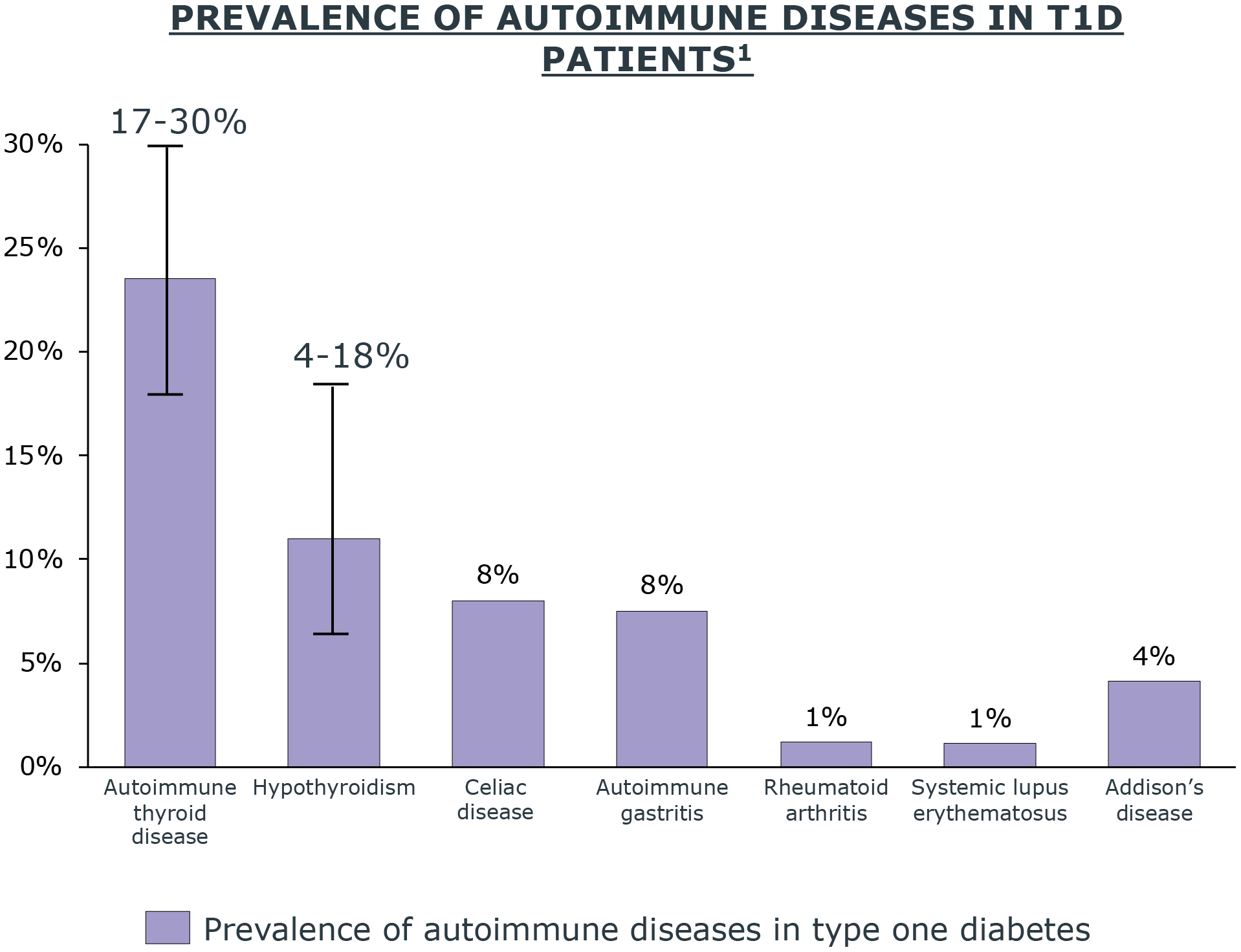
Autoimmune Diseases
- Autoimmune thyroid disorders affect more than 90% of people with T1D and autoimmune disorders11
- Around 20% of children with T1D have antithyroid antibodies present at the start of the disease11
- T1D patients often have other co-existing multi-system autoimmune disorders including thyroid, parathyroid, celiac, vitiligo, gastritis, skin, and rheumatic diseases11
Reference ongoing RWE regarding at-risk population for screening to highlight correlation between autoimmune disease and T1D
Individuals who have family antecedents with aT1D have up to 15x higher risk of developing T1D, per consensus guidelines (ADA, BRT1D, BreakthroughT1D)

The incidence and prevalence of T1D are increasing globally12
The global prevalence of T1D will rise to 13.5–17.4 million by 204013
(60–107% rise by 2040)

Only 15% of newly diagnosed people with T1D have a family history of the disease14
15-fold higher risk of developing T1D in first-degree relative15

The risk of cardiovascular complications increases the earlier the age an individual progresses to Stage 316
The prevalence of DKA at onset of aT1D is alarmingly high17
Clinical guidelines recommend early detection programs to target familial +/- autoimmune history; and acknowledge value of screening broader populations.

Recommend relatives of people with T1D be informed of the opportunity to be tested for IA in the setting of a clinical research study18
Recommendations expanded to include IA screening for anyone in the setting of a research study and screening outside of research for 1st degree family members of people with T1D19,20
These recommendations reflected the initiation of general-population screening studies and commercial availability for testing for four T1D IAs21,22

2022 guidelines acknowledge the value of screening the general population and the importance of coupling IA screening with education and monitoring
- Rewers M, et al. Presented at European Association for the Study of Diabetes 2019 Annual Meeting; Poster 279
- Ziegler AG, et al. JAMA. 2020;323:339-51
- Barker JM, et al. Diabetes Care. 2004;27:1399-404
- Larsson HE, et al. Diabetes Care. 2011;34:2347-52.
- Hummel et al. (2023) - Children diagnosed with presymptomatic type 1 diabetes through public health screening have milder diabetes at clinical manifestation
- My Child Is Islet Autoantibody Positive: Impact on Parental Anxiety - https://doi.org/10.2337/dc17-0166
- Muñoz C, Floreen A, Garey C, et al. Clin Diabetes. 2019;37(3):276-281.
- https://doi.org/10.1016/S1473-3099(24)00215-9
- https://doi.org/10.1007/s40121-021-00573-w
- https://diabetesjournals.org/diabetes/article/73/Supplement_1/1878-LB/155187/1878-LB-Medication-Use-in-Adults-with-Type-1
- Popoviciu MS, Kaka N, Sethi Y, Patel N, Chopra H, Cavalu S. Type 1 Diabetes Mellitus and Autoimmune Diseases: A Critical Review of the Association and the Application of Personalized Medicine. J Pers Med. 2023 Feb 26;13(3):422. doi: 10.3390/jpm13030422. PMID: 36983604; PMCID: PMC10056161.
- Mobasseri M, et al. Health Promot Perspect 2020;10:98–115
- Gregory GA, et al. Lancet Diabetes Endocrinol 2022;10:741–60
- JDRF. https://www.jdrf.org/blog/2023/03/17/who-is-at-risk-for-type-1-diabetes/#:~:text=Having%20a%20family%20history%20of,family%20members%20with%20the%20disease. (Accessed: 20 February 2024)
- Parkkola A, et al. Diabetes Care 2013;36:348–54
- Chiesa, ST. and ML Marcovecchio. Frontiers in Pediatrics 2021:696499
- Todd Alonso G, et al. Diabetes Care 2020;43:117–21.
- American Diabetes Association. Diabetes Care 2015;38(Suppl):S8–S16
- American Diabetes Association. Diabetes Care 2018;41(Suppl 1):S13–S27;
- American Diabetes Association. Diabetes Care 2017;40(Suppl 1):S11–S24
- Gesualdo PD, et al. Pediatr Diabetes 2016;17:441–448
- Raab J, et al. BMJ Open 2016;6:e011144
GULF:
One JLT Building, Level 3, Jumeirah Lake Towers, PO Box 53899, Dubai, UAE .
For further medical information, please contact: For UAE ✆ 800 MEDICAL Toll-Free Number.
For all Gulf countries ✆ +971 45 50 38 63 or email: medical-information.gulf@sanofi.com.
Full prescribing information is available upon request.
To Report adverse events please call: +971 561747001 or email Gulf.Pharmacovigilance@sanofi.com
www.sanofi.com
KSA:
To report any side effect(s):
Saudi Arabia: The National Pharmacovigilance and Drug Safety Centre (NPC
SFDA call center: 19999 • E-mail: npc.drug@sfda.gov.sa • Website: https://ade.sfda.gov.sa/
Full Prescribing Information is available upon request: SANOFI, Kingdom of Saudi Arabia, P.O. Box 9874, Jeddah 21423, K.S.A.
Tel: +966-12-669-3318, Fax: +966-12-663-6191
For Medical Information, please contact: +966-12-669-3318, ksa.medicalinformation@sanofi.com
For Pharmacovigilance, please contact: +966-54-428-4797, ksa_pharmacovigilance@sanofi.com
To report any Product Technical Complaint, please contact SANOFI Quality Department:
Email: quality.greatergulf@Sanofi.com , www.sanofi.com.sa
MAT-KW-2600001-V1-Feb 2026