- Article
- Source: Campus Sanofi
- Sep 18, 2025
Tzield Efficacy and Safety
Tzield is the first therapy developed & approved specifically for T1D that can delay the disease onset, and this delay translates to short term and long-term benefits
By preserving beta-cell function, Tzield significantly delays the onset of Stage 3 by median ~5 years (vs 2yrs placebo), with some subjects exceeding 7.5 years without symptoms.1
Stage 2 Study: A significant delay was observed in the Tzield treated group compared to placebo in the onset of clinical T1D
Primary analysis with median 2 years of follow-up
Median time to diagnosis of Stage 3 T1D
- Tzield: 48.4 months
- Placebo: 24.4 months
- ~39% patients were still insulin free at 5 years
- Some subjects yet to develop diabetes >7.5 years post treatment
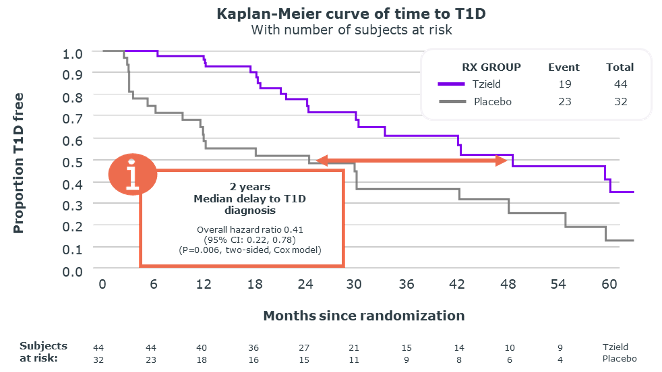
Tzield’s ability to delay onset of Stage 3 is linked to near-term improvements in quality of life and long-term morbidity and mortality benefits1
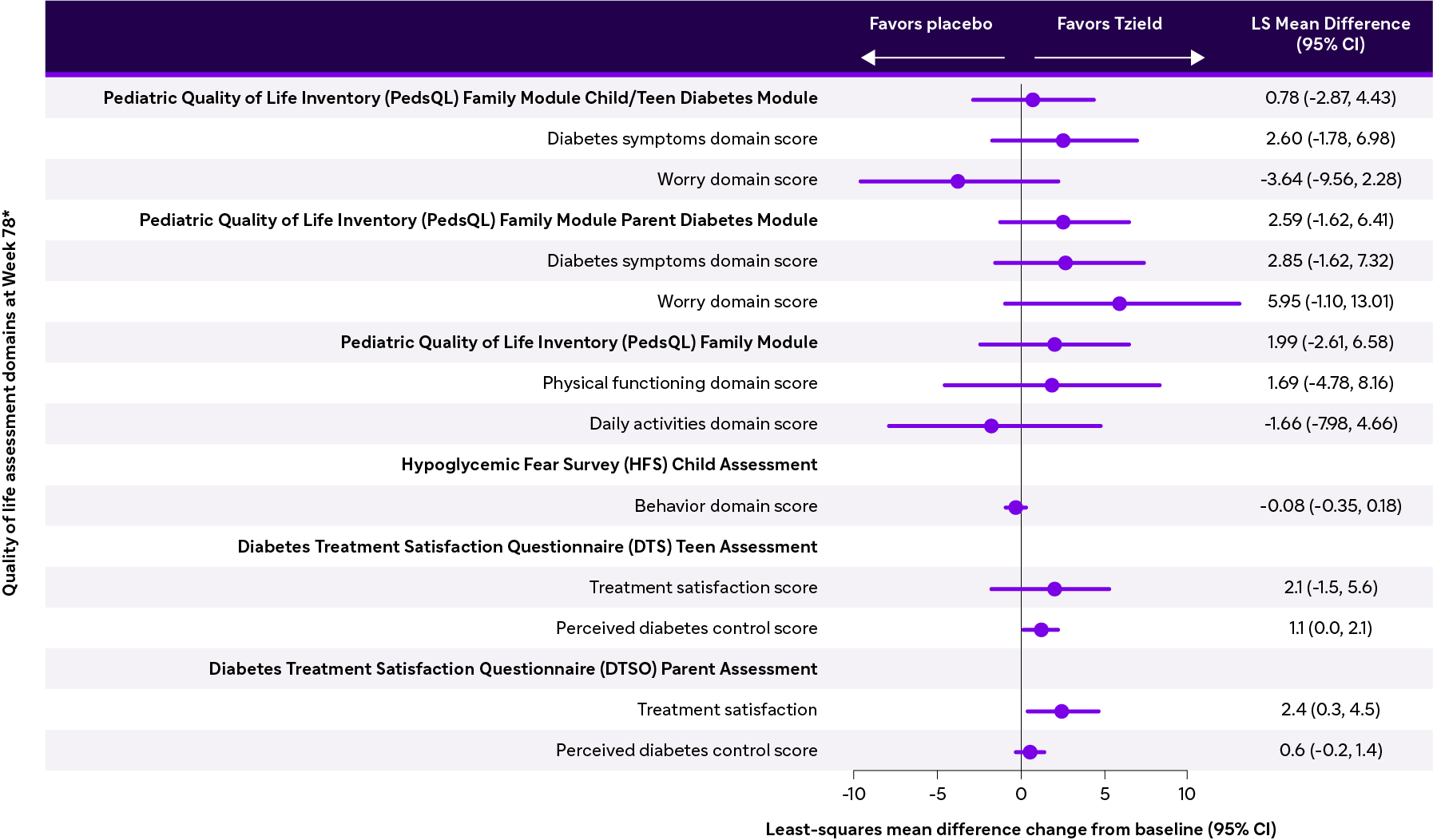
*Estimates and p-value are based on an MMRM model with treatment group, visit, age group at randomization, screening peak C-peptide category, baseline score, and a treatment by visit interaction term as fixed effects. Least-squares mean difference = Tzield – placebo. PedsQL scores range from 0 to 100 where a higher score indicates a better outcome. Minimal clinically important difference scores in PedsQL were 5.27 for child/teen and 4.54 for parents.11 HFS scores range from 0 to 4 where a lower score indicates a better outcome. Signs have been adjusted for graphical representation. DTSQ scores range from 0 to 48 where a higher score indicates a better outcome. CI, confidence interval; DTSQ, Diabetes Treatment Satisfaction Questionnaire; HFS, Hypoglycemic Fear Survey; LS, least squares; PedsQL, Pediatric Quality of Life Inventory.
Quality of Life2
Tzield demonstrated statistically significant improvement in perceived diabetes control score and treatment satisfaction via DTSQ instrument used in TN-10 study
- Perceived diabetes control scores in teens decreased from baseline to week 78 from 9.2 to 7.8 in the Tzield group and from 9.1 to 7.3 in the placebo group (LSM difference: 1.1; 95% Cl: 0.0, 2.1)
- Treatment satisfaction scores in parents increased from baseline to week 78 from 35.7 to 38.5 in the Tzield group and from 34.6 to 36.3 in the placebo groups (LSM difference: 2.4; 95% Cl: 0.3, 4.5)
The Tzield group experienced less decline in perceived diabetes control at Week 78 Parents indicated greater satisfaction with their teen's treatment at Week 78
Long-term safety data has been collected from over 1000 children & adults who have participated in clinical trials since 2001. Adverse events typically resolve over a short period of time.1,2
- Safety was consistent with previous experience; no new safety signals
- The most common adverse events leading to study drug discontinuation were protocol defined LFT elevations (Tzield 6.9%; placebo 2.7%) and cytokine release syndrome (CRS) (Tzield 1.8%; placebo 0%)
- Similar rates of SAEs observed in Tzield 5.5% (CRS 1.4%) and placebo 5.4% (infections 2.7%)
- EBV reactivation was detected in 8 Tzield treated patients and was asymptomatic in 6; there was one new case of EBV in each treatment group.
All events resolved without antiviral treatment
| Tzield (n = 217) | Placebo (n = 111) | |
|---|---|---|
| Hypoglycemia |
151 (69.6) |
81 (73.0) |
| Headache |
94 (43.3) |
21 (18.9) |
| Nausea |
92 (42.4) |
21 (18.9) |
| Rash |
86 (39.6) |
5 (4.5) |
| Lymphocyte count decreased |
73 (33.6) |
5 (4.5) |
| Vomiting |
69 (31.8) |
15 (13.5) |
Safety data from 791 teplizumab-treated participants (children & adults) across Stage 2 and Stage 3 autoimmune T1D clinical trials show that teplizumab has a favorable safety profile characterized by self-limited AEs.15
- Most AEs were Grade 1 (mild) or 2 (moderate) in severity and typically occurred during or immediately following the dosing period and resolved without intervention.15
- AEs leading to permanent discontinuation of teplizumab were reported in 14.3% and 3.7% of participants in the teplizumab and control groups, respectively, across five clinical trials.15
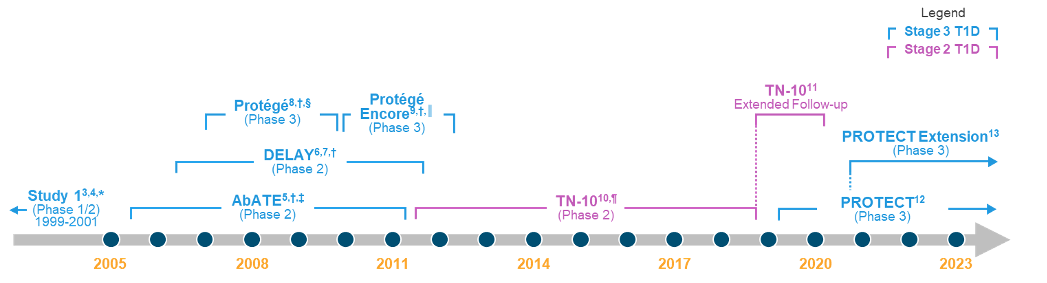
*Data from study included in pooled efficacy analysis.14
†Data from study included in pooled efficacy and safety analyses.12
‡Study period was 2 years. A 7-year follow-up study was conducted at the conclusion of the clinical trial.5,15
§Terminated early due to futility on its primary endpoint.15
║Terminated prematurely with 254 subjects (64% of the planned 400 subjects) enrolled after Protégé failed to demonstrate efficacy on the primary endpoint.15
¶Data from study included in pooled safety analysis.14
These data from 791 teplizumab-treated individuals represent 1500 patient-years of follow-up, the most extensive dataset in autoimmune T1D disease modification with a single agent to date.15
By preserving beta-cell function, Tzield significantly delays the onset of Stage 3 by median ~5 years (vs 2yrs placebo), with some subjects exceeding 7.5 years without symptoms.1
Stage 2 Study: A significant delay was observed in the Tzield treated group compared to placebo in the onset of clinical T1D
Primary analysis with median 2 years of follow-up
Median time to diagnosis of Stage 3 T1D
- Tzield: 48.4 months
- Placebo: 24.4 months
- ~39% patients were still insulin free at 5 years
- Some subjects yet to develop diabetes >7.5 years post treatment

Tzield’s ability to delay onset of Stage 3 is linked to near-term improvements in quality of life and long-term morbidity and mortality benefits1

*Estimates and p-value are based on an MMRM model with treatment group, visit, age group at randomization, screening peak C-peptide category, baseline score, and a treatment by visit interaction term as fixed effects. Least-squares mean difference = Tzield – placebo. PedsQL scores range from 0 to 100 where a higher score indicates a better outcome. Minimal clinically important difference scores in PedsQL were 5.27 for child/teen and 4.54 for parents.11 HFS scores range from 0 to 4 where a lower score indicates a better outcome. Signs have been adjusted for graphical representation. DTSQ scores range from 0 to 48 where a higher score indicates a better outcome. CI, confidence interval; DTSQ, Diabetes Treatment Satisfaction Questionnaire; HFS, Hypoglycemic Fear Survey; LS, least squares; PedsQL, Pediatric Quality of Life Inventory.
Quality of Life2
Tzield demonstrated statistically significant improvement in perceived diabetes control score and treatment satisfaction via DTSQ instrument used in TN-10 study
- Perceived diabetes control scores in teens decreased from baseline to week 78 from 9.2 to 7.8 in the Tzield group and from 9.1 to 7.3 in the placebo group (LSM difference: 1.1; 95% Cl: 0.0, 2.1)
- Treatment satisfaction scores in parents increased from baseline to week 78 from 35.7 to 38.5 in the Tzield group and from 34.6 to 36.3 in the placebo groups (LSM difference: 2.4; 95% Cl: 0.3, 4.5)
The Tzield group experienced less decline in perceived diabetes control at Week 78 Parents indicated greater satisfaction with their teen's treatment at Week 78
Long-term safety data has been collected from over 1000 children & adults who have participated in clinical trials since 2001. Adverse events typically resolve over a short period of time.1,2
- Safety was consistent with previous experience; no new safety signals
- The most common adverse events leading to study drug discontinuation were protocol defined LFT elevations (Tzield 6.9%; placebo 2.7%) and cytokine release syndrome (CRS) (Tzield 1.8%; placebo 0%)
- Similar rates of SAEs observed in Tzield 5.5% (CRS 1.4%) and placebo 5.4% (infections 2.7%)
- EBV reactivation was detected in 8 Tzield treated patients and was asymptomatic in 6; there was one new case of EBV in each treatment group.
All events resolved without antiviral treatment
| Tzield (n = 217) | Placebo (n = 111) | |
|---|---|---|
| Hypoglycemia |
151 (69.6) |
81 (73.0) |
| Headache |
94 (43.3) |
21 (18.9) |
| Nausea |
92 (42.4) |
21 (18.9) |
| Rash |
86 (39.6) |
5 (4.5) |
| Lymphocyte count decreased |
73 (33.6) |
5 (4.5) |
| Vomiting |
69 (31.8) |
15 (13.5) |
Safety data from 791 teplizumab-treated participants (children & adults) across Stage 2 and Stage 3 autoimmune T1D clinical trials show that teplizumab has a favorable safety profile characterized by self-limited AEs.15
- Most AEs were Grade 1 (mild) or 2 (moderate) in severity and typically occurred during or immediately following the dosing period and resolved without intervention.15
- AEs leading to permanent discontinuation of teplizumab were reported in 14.3% and 3.7% of participants in the teplizumab and control groups, respectively, across five clinical trials.15

*Data from study included in pooled efficacy analysis.14
†Data from study included in pooled efficacy and safety analyses.12
‡Study period was 2 years. A 7-year follow-up study was conducted at the conclusion of the clinical trial.5,15
§Terminated early due to futility on its primary endpoint.15
║Terminated prematurely with 254 subjects (64% of the planned 400 subjects) enrolled after Protégé failed to demonstrate efficacy on the primary endpoint.15
¶Data from study included in pooled safety analysis.14
These data from 791 teplizumab-treated individuals represent 1500 patient-years of follow-up, the most extensive dataset in autoimmune T1D disease modification with a single agent to date.15
- Herold KC, et al. N Engl J Med. 2019;381(7):603-13.
- Ramos EL, et al. N Engl J Med 2023;389:2151–61.
- Herold KC, et al. N Engl J Med. 2002;346(22):1692-1698.
- Herold KC, et al. Diabetes. 2005;54(6):1763-1769.
- Herold KC, et al. Diabetes. 2013;62(11):3766-3774.
- Herold KC, et al. Diabetologia. 2013;56(2):391-400.
- ClinicalTrials.gov. NCT00378508. Updated August 13, 2021. Accessed March 22, 2023. https://clinicaltrials.gov/ct2/show/NCT00378508
- Sherry N, et al. Lancet. 2011;378(9790):487-497.
- ClinicalTrials.gov. NCT00920582. Updated February 8, 2022. Accessed March 19, 2023. https://clinicaltrials.gov/ct2/show/NCT00920582
- Herold KC, et al. N Engl J Med. 2019;381(7):603-613.
- Sims EK, et al. Sci Transl Med. 2021;13(583):eabc8980.
- ClinicalTrials.gov. NCT03875729. Updated February 9, 2023. Accessed March 19, 2023. https://clinicaltrials.gov/ct2/show/NCT03875729
- ClinicalTrials.gov. NCT04598893. Updated February 9, 2023. Accessed March 30, 2023. https://clinicaltrials.gov/ct2/show/NCT04598893
- Data on file. Provention Bio, Inc. 13. Perdigoto AL, et al. Diabetologia. 2019;62(4):655-664. 14. US Food and Drug Administration. Endocrinologic and Metabolic Drugs Advisory Committee Meeting. May 27, 2021. Accessed March 3, 2023. https://www.fda.gov/media/149388/download
- Herold KC, et al. Diabetes Care. 2023;46(10):1848-56 and supplement [Stage 3 autoimmune T1D].
GULF:
One JLT Building, Level 3, Jumeirah Lake Towers, PO Box 53899, Dubai, UAE .
For further medical information, please contact: For UAE ✆ 800 MEDICAL Toll-Free Number.
For all Gulf countries ✆ +971 45 50 38 63 or email: medical-information.gulf@sanofi.com.
Full prescribing information is available upon request.
To Report adverse events please call: +971 561747001 or email Gulf.Pharmacovigilance@sanofi.com
www.sanofi.com
KSA:
To report any side effect(s):
Saudi Arabia: The National Pharmacovigilance and Drug Safety Centre (NPC
SFDA call center: 19999 • E-mail: npc.drug@sfda.gov.sa • Website: https://ade.sfda.gov.sa/
Full Prescribing Information is available upon request: SANOFI, Kingdom of Saudi Arabia, P.O. Box 9874, Jeddah 21423, K.S.A.
Tel: +966-12-669-3318, Fax: +966-12-663-6191
For Medical Information, please contact: +966-12-669-3318, ksa.medicalinformation@sanofi.com
For Pharmacovigilance, please contact: +966-54-428-4797, ksa_pharmacovigilance@sanofi.com
To report any Product Technical Complaint, please contact SANOFI Quality Department:
Email: quality.greatergulf@Sanofi.com , www.sanofi.com.sa
MAT-KW-2600001-V1-Feb 2026