- Article
- Source: Campus Sanofi
- Sep 18, 2025
Tzield MOA and Dosing
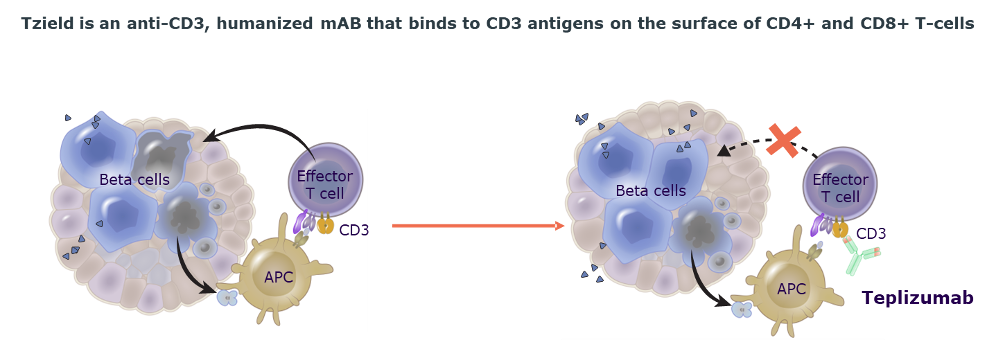
Tzield is the first and only disease modifying immunotherapy developed for aT1D that treats the underlying cause of the disease by preserving beta-cell function.1-3
Mechanism of Action
Prescription and Dosing
Unlike other chronic diabetes therapies, Tzield is a finite course of treatment. For Stage 2,
Tzield is administered through a single cycle of 14-day IV infusion.5
- Teplizumab is administered by IV (over a minimum of 30 minutes) once daily for 14 consecutive days
- The recommended dose for adults and pediatric patients aged ≥8 years is based on BSA according to the following:
- Two doses should not be administered on the same day
- If a planned teplizumab infusion is missed, resume dosing by administering all remaining doses on consecutive days to complete the 14-day treatment course
- Kuhn and Weiner, 2016;
- Chen and Flies, 2013.
- Murphy K, Weaver C. Janeway’s Immunobiology. 9th ed. Garland Science; 2017:257-93. Kuhn C, Weiner HL. Immunotherapy. 2016;8(8):889-906; 3. Long A, et al. Sci Immunol, 2016;1:eaai7793.
- Herold KC, et al. N Engl J Med 2019;381:603–13.
- TZIELD. Prescribing Information. Provention Bio, Inc
MAT-KW-2600001-V1-Feb 2026