- Article
- Source: Campus Sanofi
- 11 Mar 2026
Improving Quality of Life in Patients with Uncontrolled Atopic Dermatitis (AD): Tools for Timely Identification and Treatment
DUPIXENT® (dupilumab) Prescribing Information UK
Introduction
AD, a chronic inflammatory skin condition, poses significant challenges in paediatric, adolescent and adult patients.1 A key factor in the pathophysiology of AD is the underlying type 2 inflammation, driven by cytokines such as IL-4 and IL-13.2,3 This type 2 inflammation can contribute to barrier dysfunction, intense itch and increased risk of skin infections.2,3 The impact of moderate-to-severe AD is multifaceted, and the long-term risks of uncontrolled AD may extend beyond visible skin symptoms.4,5
Understanding the Burden Beyond Skin Symptoms
The symptoms of AD can affect not only the skin but also the overall QoL, including social, emotional, and physical health. The combined effect of these factors, over time, in uncontrolled AD can lead to cumulative life course impairment (CLCI) for some patients.4–7 Recognising these broader implications is essential for an effective management.6
A range of tools have been developed to help identify those patients whose AD remains uncontrolled despite current therapy.6,8
Tools for Identifying Uncontrolled AD
The Atopic Dermatitis Control Tool (ADCT) provides valuable insights into evaluating the extent of the condition and guiding healthcare providers to make informed treatment decisions.9,10
Atopic Dermatitis Control Tool (ADCT): This patient-reported tool is designed to facilitate meaningful discussions between patients and healthcare providers regarding the control of AD in clinical practice. This validated tool is appropriate for patients 12 years and older and it consists of six questions that evaluate the severity of symptoms, including itch and sleep disturbances, and their impact on daily activities and mood. Within a range of 0 to 24, a score of ≥7 indicates inadequate disease control, which prompts a reassessment of treatment strategies. The ADCT helps in identifying the degree of disease control and making informed decisions about escalating therapy.9,10
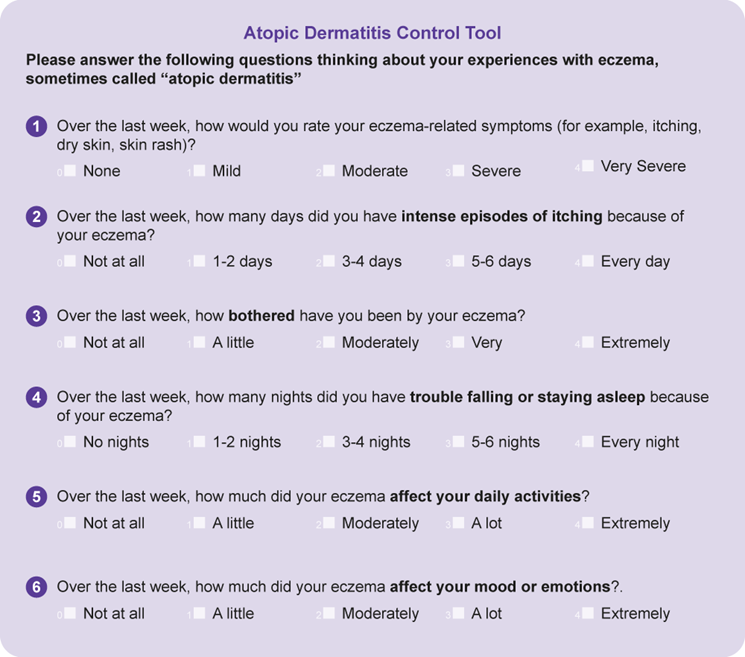
Please Click here to access the ADCT – a validated, brief and easily scored patient self-assessment tool.
Potential Future Tools for Assessing Cumulative Life Course Impairment:
Tools to address the CLCI caused by AD will be essential for understanding and managing the profound impacts of AD throughout patients’ lives. Recent developments in this area show promise, though it's important to note these tools have not been validated and are still in development stages for AD (and other chronic skin conditions).8
Dermatology CLCI Retrospective (DermCLCI-r) tool
Aims to assess the CLCI caused by AD, focusing on how the disease has affected patients’ lives over time. It includes questions on physical, emotional, and social domains, offering a comprehensive view of past impacts.8

Dermatology CLCI prospective (DermCLCI-p) tool
Conversely, the DermCLCI-p tool is intended to predict future risks associated with AD, helping to identify patients at risk for life impairment.8
Both tools consist of 30-item questionnaire that helps assess the impairments of AD. This questionnaire assesses the following:8
- The effect of life course impairment on a 4-point Likert scale [0 (not at all) to 3 (very)] and whether these have been life-changing
- The effect of coping behaviours on a 5-point Likert scale [0 (not at all) to 4 (very)]
These tools are still in their early stages and await comprehensive psychometric testing for validity and reliability. Development of these tools is an important first step in better understanding the experience of disease burden within the context of additional impactful life events. With future study, they hold promise for integration into both routine clinical care and broader health services research.8
Using tools to identify where patients' AD is inadequately controlled could help inform treatment strategies. When AD is refractory to topical therapies, advanced treatment with phototherapy or systemic medications may be considered. Advanced systemic therapies, including biologics or JAKis, are considered effective in treating eligible patients with moderate-to-severe AD.11–14
Conclusion
Understanding and using these tools can contribute to better management of AD.10 By recognising the extensive burden of AD beyond the skin, clinicians are better equipped to identify patients who are not well-controlled on their current medications. This awareness may better inform decisions about when treatment escalation may be required to address both immediate symptoms and long-term impact on the patient’s QoL.8,10
Early identification of at-risk patients, using appropriate tools, and timely escalation to appropriate treatments are crucial in providing best practice care for patients.15–17
Discover tools that can help you identify AD patients that are uncontrolled.
DUPIXENT is approved for use in eligible patients as young as 6 months old in AD.18
DUPIXENT is indicated for use in patients who are candidates for systemic therapy, aged 12 years and older with moderate-to-severe atopic dermatitis (AD), and in patients aged 6 months to 11 years of age with severe AD.18
AD, atopic dermatitis; ADCT, Atopic Dermatitis Control Test; CLCI, Cumulative Life Course Impairment; IL, interleukin; JAKis, Janus kinase inhibitors; QoL, quality of life.
References
- Weidinger S et al. Lancet. 2016;387(10023):1109–1122.
- Haddad EB et al. Dermatol Ther (Heidelb). 2022;12(7):1501–1533.
- Moniaga CS et al. Diagnostics. 2021; 11(11):2090.
- Fasseeh AN et al. Dermatol Ther (Heidelb). 2022;12(12):2653–2668.
- Weidinger S et al. Br J Dermatol. 2024;190(6):846–857.
- Calzavara-Pinton P et al. Adv Ther. 2023;40(12):5366–5382.
- Augustin M et al. JEADV. 2022; 36(7):3–16.
- Braren-von Stülpnagel CC et al. J Eur Acad Dermatol Venereol. Published online February 18, 2023. doi:10.1111/jdv.18977
- Strober B et al. JAMA Dermatol. 2022;158(2):142–150.
- ADCT, Atopic Dermatitis Control Tool. Patient Form. https://www.adcontroltool.com/adct-downloads1 Date last accessed: March 2026.
- Chovatiya R et al. Am J Clin Dermatol. 2022;23(4):459–468.
- Davis DMR et al. J Am Acad Dermatol. 2024;90(2):e43–e56.
- Chu DK et al. Ann Allergy Asthma Immunol. 2024;132(3):274–312.
- Chu AWL et al. J Allergy Clin Immunol. 2023;152(6):1470–1492.
- Augustin M, et al. JEADV. 2022; 36(7): 3–16.
- von Stülpnagel CC et al. J Eur Acad Dermatol Venereol. 2021; 35(11): 2166–2184.
- Bieber T. Nat Rev Drug Discov. 2023; 22(8): 662–680.
- DUPIXENT Summary of Product Characteristics. 2026.

Atopic Dermatitis Control Test (ADCT)
Atopic dermatitis, a type of eczema, may be affecting your patient’s life in more ways than you know.
The ADCT gives a measure of how controlled your patient’s eczema is. Use these 6 concise questions to evaluate all dimensions of atopic dermatitis control.
Development of ADCT involved literature review as well as interviews with patients and physicians, and was funded by Sanofi and Regeneron.
Try the ADCTMAT-XU-2600675 (v1.0) Date of Preparation: March 2026