- Article
- Source: Campus Sanofi
- 23 Feb 2026
About Beyfortus®▼ (Nirsevimab): Clinical Efficacy, Real-World Impact, and Duration of Protection
Beyfortus is the first RSV immunisation designed to offer protection to all infants, with effectiveness and public health impact supported by real-world data1-5
Watch Professor Saul N Faust and Dr Katrina Cathie discuss Beyfortus in a series of three videos

Professor Saul N Faust
Professor of Paediatric Immunology & Infectious Diseases
NIHR Southampton Clinical Research Facility and Biomedical Research Centre, University Hospital Southampton NHS Foundation Trust

Dr Katrina Cathie
Consultant Paediatrician & Associate Director,
NIHR Southampton Clinical Research Facility and University Hospital Southampton NHS Foundation Trust
Direct
Offers direct protection against RSV LRTD via administration of antibodies without relying on an infant's immune system1,6
Rapid
Offers RSV protective antibodies starting Day 1 after injection, reaching maximum concentration within 6 days (range 1 to 28 days)1*
Durable
Beyfortus is a long-acting monoclonal antibody modified to extend the half-life to ~71 days1†
A single dose offers protection for at least 5 to 6 months1
*Following intramuscular administration, maximum concentration was reached within 6 days (range 1‑28 days).1
†Beyfortus has been modified with a triple amino acid substitution (YTE) in the Fc region to extend serum half-life.1
Beyfortus reduced the risk of medically attended RSV LRTI and has demonstrated a reduction in RSV hospitalisation versus placebo7,8
Beyfortus has been studied in a robust clinical trial program across a broad infant population, including those born:7-9
- term or pre-term.
- healthy, or with underlying health conditions (including chronic lung disease of prematurity (CLD), congenital heart disease (CHD)).
- during or before the RSV season.
≤35 weeks gestation without CHD or CLD, n=615; infants with uncorrected, partially corrected or medically treated CHD or CLD, n=310. The study evaluated the safety profile and pharmacokinetics of Beyfortus. The incidence of adverse events was similar between Beyfortus and palivizumab and at Day 151 serum Beyfortus levels were similar in both cohorts.9
≥29 to ≤32 weeks gestation, n=363; >32 weeks gestation, n=606. Primary endpoint was medically attended RSV-associated LRTI through 150 days after dose administration, which was met.7
≥35 to <37 weeks gestation, n=132; ≥37 weeks gestation, n=861. Primary endpoint was medically attended RSV-associated LRTI through 150 days after injection, which was met.8
Watch Professor Saul N Faust and Dr Katrina Cathie present the efficacy and safety profile of Beyfortus
Time to watch: 7 mins 4 secs

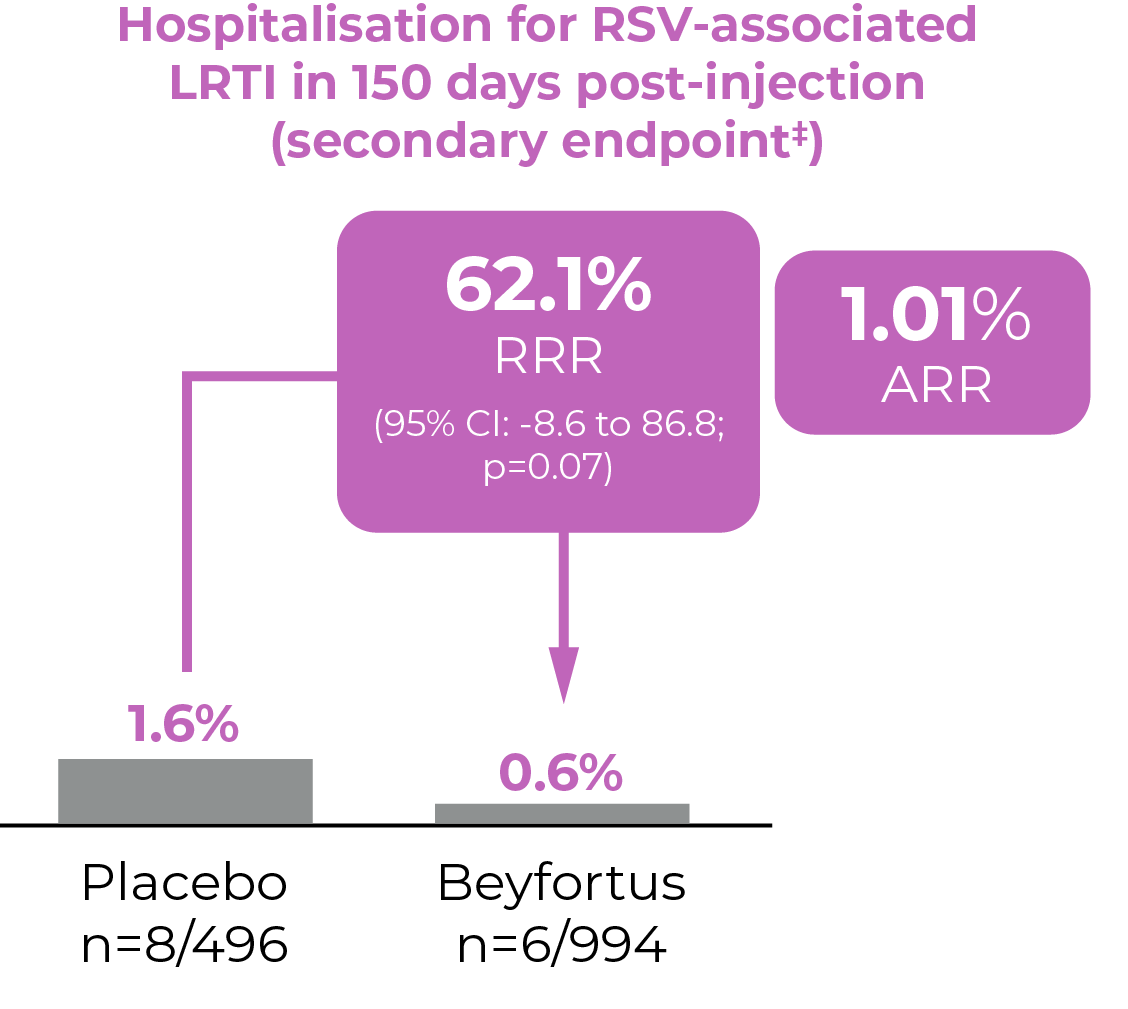
Adapted from Hammitt L L et al, 20228
†Primary endpoint was met
‡Secondary endpoint did not meet statistical significance
Adverse events of grade 3 or higher severity were reported in 36 of the 987 infants (3.6%) who received Beyfortus and in 21 of the 491 infants (4.3%) who received placebo.8
A single dose of Beyfortus for an entire RSV season1,2,8§
A single dose of Beyfortus offers protection against RSV LRTI for at least 5-6 months, covering the duration of a typical RSV season.1,2,8§
For complete prescribing information, refer to the Summary of Product Characteristics.
§Weight-based dosing regimen.1
Watch Professor Saul N Faust and Dr Katrina Cathie discuss the duration of protection and dosage of Beyfortus
Time to watch: 3 mins 49 secs
Real‑world evidence for a single dose of Beyfortus3,10
High effectiveness (versus no immunisation) and public health impact was consistently seen with Beyfortus in real‑world studies, supporting its use as an effective RSV immunisation.10,11
Watch Professor Saul N Faust and Dr Katrina Cathie further explore real-world evidence for Beyfortus
Time to watch: 4 mins 40 secs
NIRSE-GAL:3 hospitalisations for RSV-associated LRTI (catch-up cohort)‖
Effectiveness
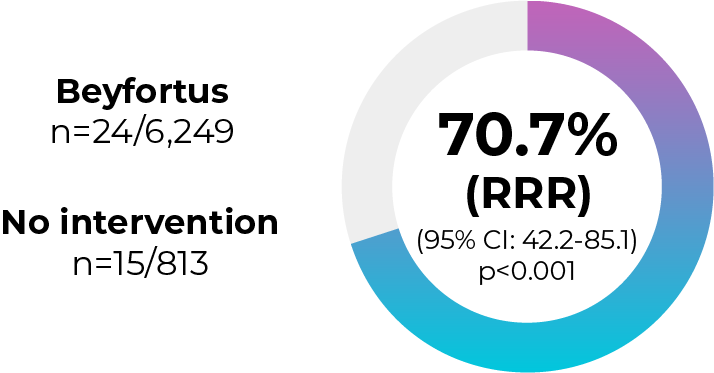
0.386% ARR per 10,000 person-days
Intention‑to‑treat analysis (n=7,061) using catch-up group only; Beyfortus 24 events per 1,168,165 person‑days versus 15 events per 254,019 person‑days for non‑Beyfortus recipients
NIRSE-GAL was a longitudinal, multicentre, population-based observational study in infants 0-24 months (n=10,259) performed in Spain during the 2023‑2024 RSV season. Immunisation uptake was over 90% overall. The primary endpoint was hospital admissions for RSV-related LRTI, which was met.11
No severe adverse events directly related to Beyfortus were registered in this study.11
The results from real‑world data analyses are limited by potential selection bias, as treatment groups often show differences in baseline characteristics. Outcome definitions may differ between RCTs and RWD analyses, and clinical events cannot be adjudicated. Certain clinical parameters may not be present in observational data; these analyses are subject to limitations.
‖Since Beyfortus uptake exceeded 90% in the seasonal cohort and combined seasonal and catch-up cohort, Cox regression models were used to estimate effectiveness from the catch-up cohort only (data shown from the intention to treat population).3
The most frequent adverse reaction was rash (0.7%) occurring within 14 days post dose. The majority of cases were mild to moderate in intensity. Additionally, pyrexia and injection site reactions were reported at a rate of 0.5% and 0.3% within 7 days post dose, respectively. Injection site reactions were non-serious.1
ARR: absolute risk reduction; CI: confidence interval; ICU: intensive care unit; IQR: interquartile range; LRTI: lower respiratory tract infection; RCT: randomised controlled trial; RRR: relative risk reduction; RWD: real‑world data
- Beyfortus UK Summary of Product Characteristics. Available at: medicines.org.uk. Accessed February 2026
- Drysdale S B, Cathie K et al. Nirsevimab for prevention of hospitalizations due to RSV in infants. N Engl J Med 2023;389:2425‑2435
- Mallah N, Pardo‑Seco J et al. Full 2023‑24 season results of universal prophylaxis with nirsevimab in Galicia, Spain: the NIRSE‑GAL study. Lancet Infect Dis 2025;25:e62‑e63
- Sumsuzzman D M, Wang Z et al. Real‑world effectiveness of nirsevimab against respiratory syncytial virus disease in infants: a systematic review and meta‑analysis. Lancet Child Adolesc Health 2025;9:393‑403
- Torres J P, Saure D et al. Effectiveness and impact of nirsevimab in Chile during the first season of a national immunisation strategy against RSV (NIRSE‑CL): a retrospective observational study. Lancet Infect Dis 2025:S1473‑3099
- Verwey C, Madhi S A. Review and update of active and passive immunization against respiratory syncytial virus. BioDrugs 2023;37:295‑309
- Griffin M P, Yuan Y et al. Single‑dose nirsevimab for prevention of RSV in preterm infants. N Engl J Med 2020;383:415‑425
- Hammitt L L, Dagan R et al. Nirsevimab for prevention of RSV in healthy late‑preterm and term infants. N Engl J Med 2022;386:837‑846
- Domachowske J, Madhi S A et al. Safety of nirsevimab for RSV in infants with heart or lung disease or prematurity. N Engl J Med 2022;386:892‑894
- Paris L, Comegan, L et al. Protecting infants from respiratory syncytial virus (RSV) in Ireland: impact of a national nirsevimab immunisation programme, 2024/2025. Preprint. Available at: papers.ssrn.com. Accessed February 2026
- Ares‑Gomez S, Mallah N et al. Effectiveness and impact of universal prophylaxis with nirsevimab in infants against hospitalisation for respiratory syncytial virus in Galicia, Spain: initial results of a population‑based longitudinal study. Lancet Infect Dis 2024;24:817‑828
MAT-XU-2600537 (v1.0) March 2026