- Article
- Source: Campus Sanofi
- 30 Mar 2026
The BRIGHT Study: Comparing Toujeo® (insulin glargine 300 U/mL) and Insulin Degludec 100 U/mL

Toujeo® (insulin glargine 300 units/ml) Prescribing Information
Effective Type 2 diabetes management involves balancing efficacy and safety considerations. When prescribing basal insulin (BI), treatment options may be associated with different glycaemic outcomes.1 The BRIGHT study provides valuable head-to-head data comparing Toujeo® (insulin glargine 300 U/mL) and insulin degludec 100 U/mL.1
BRIGHT Study: Direct Comparison
The BRIGHT trial is a randomised controlled study designed to directly compare Toujeo® (insulin glargine 300 U/mL) with degludec 100 U/mL in insulin-naïve adults with type 2 diabetes (T2D).1 This 24-week, multicentre study enrolled 929 participants.1
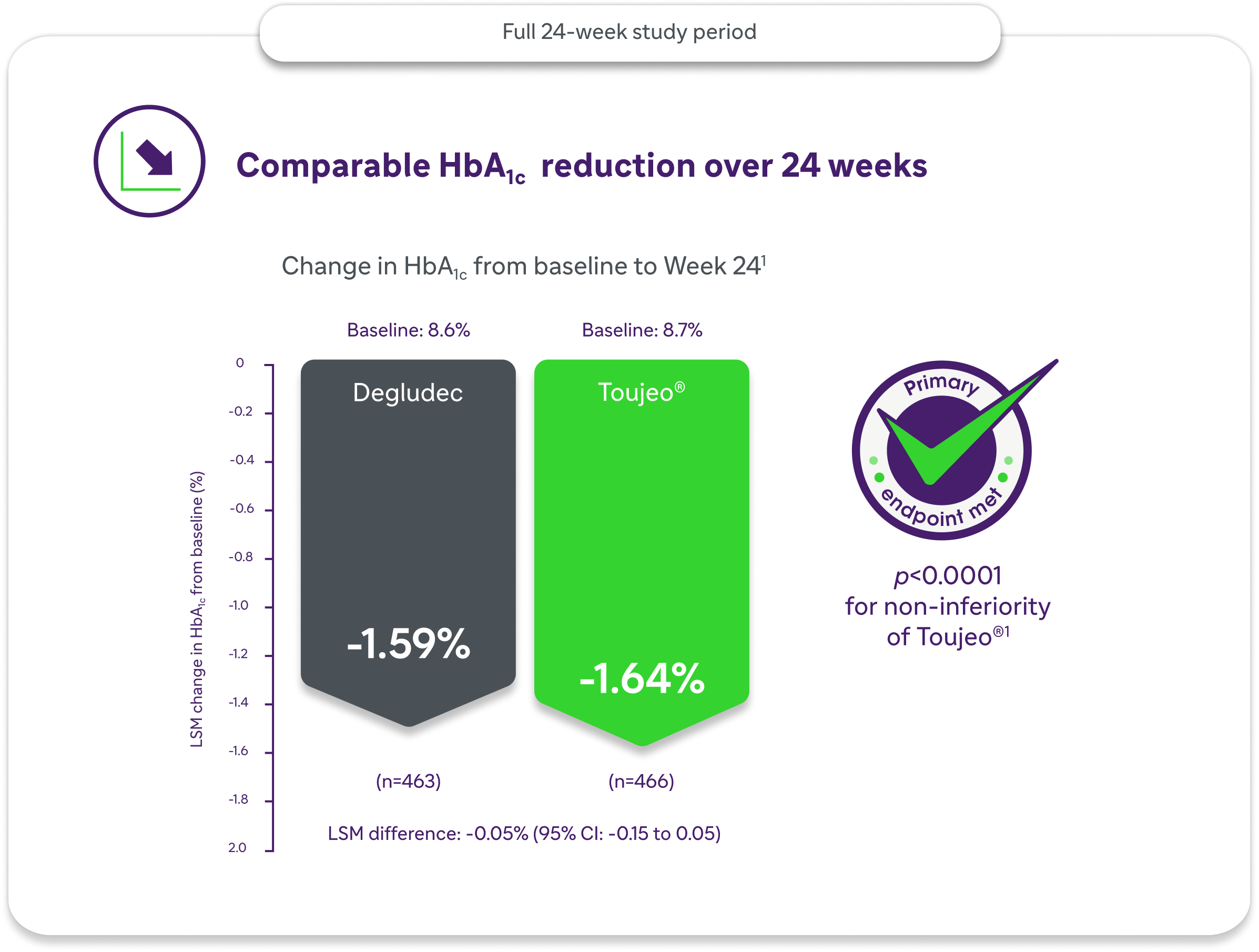
Comparable Glycaemic Efficacy: HbA1c Outcomes at Week 24
Both Toujeo® and insulin degludec 100 U/mL were associated with reductions in HbA1c over 24 weeks. Toujeo® demonstrated non-inferiority to insulin degludec 100 U/mL for the primary efficacy endpoint.1
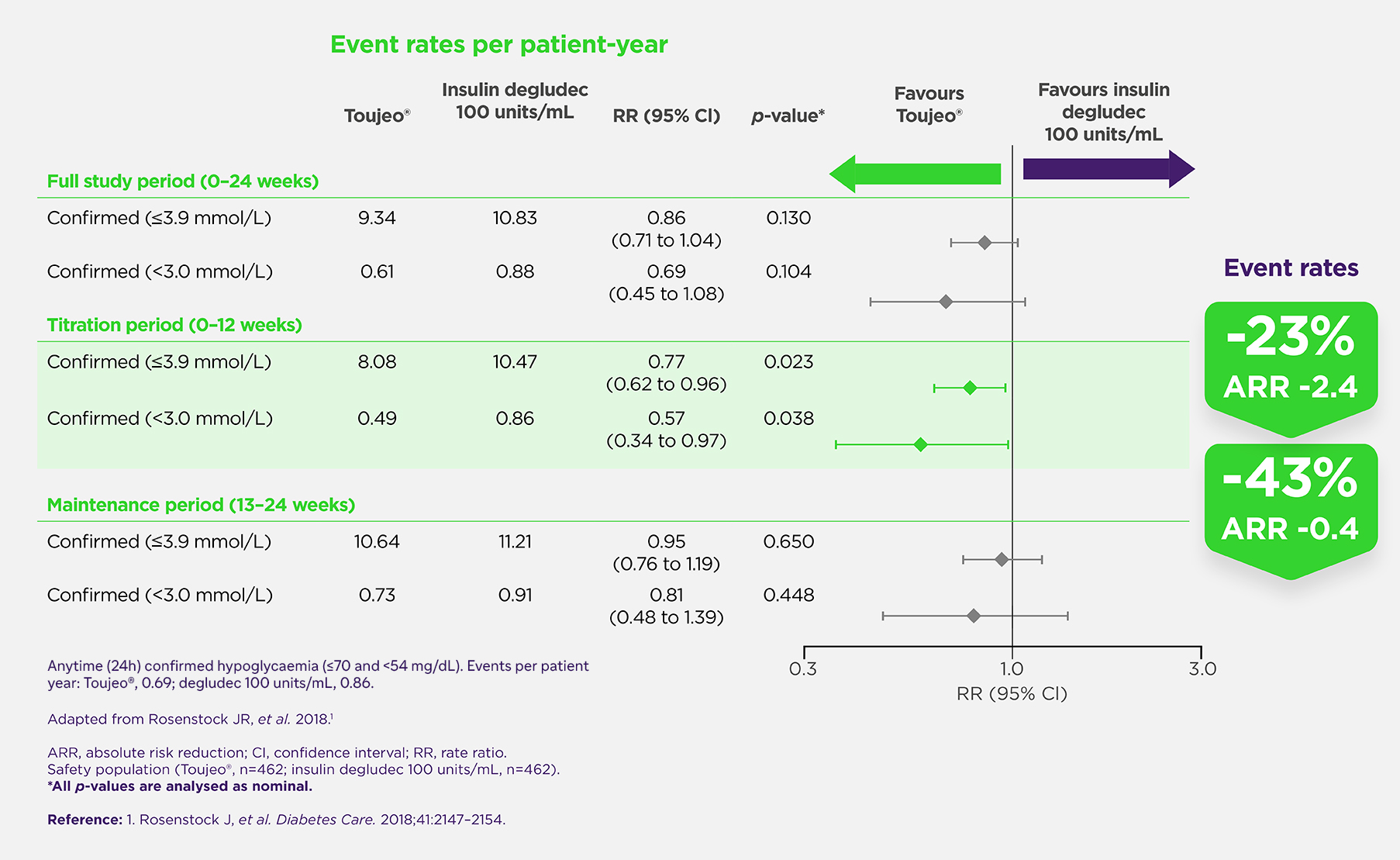
Hypoglycaemia Rates
Over the full 24-week study period, the incidence of anytime (24-h) severe and/or documented hypoglycaemia (<3.0 mmol/L) was comparable between Toujeo® and insulin degludec 100 U/mL (RR (95% CI): 0.69 (0.45 to 1.08); p=0.104)¹ During the initial 12-week titration period, hypoglycaemia outcomes were evaluated separately for Toujeo® and insulin degludec 100 U/mL and showed lower confirmed rates of anytime (24-h) severe and/or documented hypoglycaemia (<3.0 mmol/L) with Toujeo® (RR (95% CI): 0.57 (0.34 to 0.97); p=0.038).1
Conclusion
The initial weeks of basal insulin therapy represent a crucial window that may influence long-term treatment success.2 People with diabetes who experienced hypoglycaemia during the first months of insulin therapy were more likely to experience recurrent episodes and had a higher risk of hypoglycaemia and basal insulin discontinuation.2,3 Strategies to reduce hypoglycaemia risk during titration may help influence subsequent insulin dosing behaviour and longer-term safety outcomes.2,3
In this randomised study, the BRIGHT trial evaluated glycaemic and hypoglycaemic outcomes in a head-to-head comparison of Toujeo® (insulin glargine 300U/mL) and insulin degludec 100 U/mL in insulin-naïve patients with type 2 diabetes. Over the 24-week study period, non-inferiority for the primary HbA1c endpoint was demonstrated, with additional analyses describing hypoglycaemia outcomes during the titration and maintenance phases.1,3
Abbreviations
ARR, absolute risk reduction; CI, confidence interval; HbA1c, glycated haemoglobin; RR, rate ratio; T2D, type 2 diabetes
Explore More
References
- Rosenstock J, Cheng A, Ritzel R, et al. More Similarities Than Differences Testing Insulin Glargine 300 Units/mL Versus Insulin Degludec 100 Units/mL in Insulin-Naive Type 2 Diabetes: The Randomized Head-to-Head BRIGHT Trial. Diabetes Care. 2018 Oct;41(10):2147- 2154.
- Mauricio D, Meneghini L, Seufert J, et al. Glycaemic control and hypoglycaemia burden in patients with type 2 diabetes initiating basal insulin in Europe and the USA. Diabetes Obes Metab. 2017 Aug;19(8):1155-1164.
- Cheng A, Harris S, Giorgino F, et al. Similar glycaemic control and less hypoglycaemia during active titration after insulin initiation with glargine 300 units/mL and degludec 100 units/mL: A subanalysis of the BRIGHT study. Diabetes Obes Metab. 2020 Mar;22(3):346-354.
MAT-XU-2600250-1.0-03/2026
