Dosing
Indication
DUPIXENT is indicated in adults as add-on maintenance treatment for uncontrolled COPD characterised by raised blood eosinophils on a combination of an ICS, a LABA, and a LAMA, or on a combination of a LABA and a LAMA if ICS is not appropriate.1
Please refer to the Summary of Product Characteristics for further information.1
The FLEXIBILITY of at-home or in-clinic administration
DUPIXENT offers a fixed dosing schedule with no loading dose1

a300 mg of dupilumab in 2 mL solution.1
Two devices to provide confident subcutaneous administration to your patients
DUPIXENT Pre-Filled Pen2,3
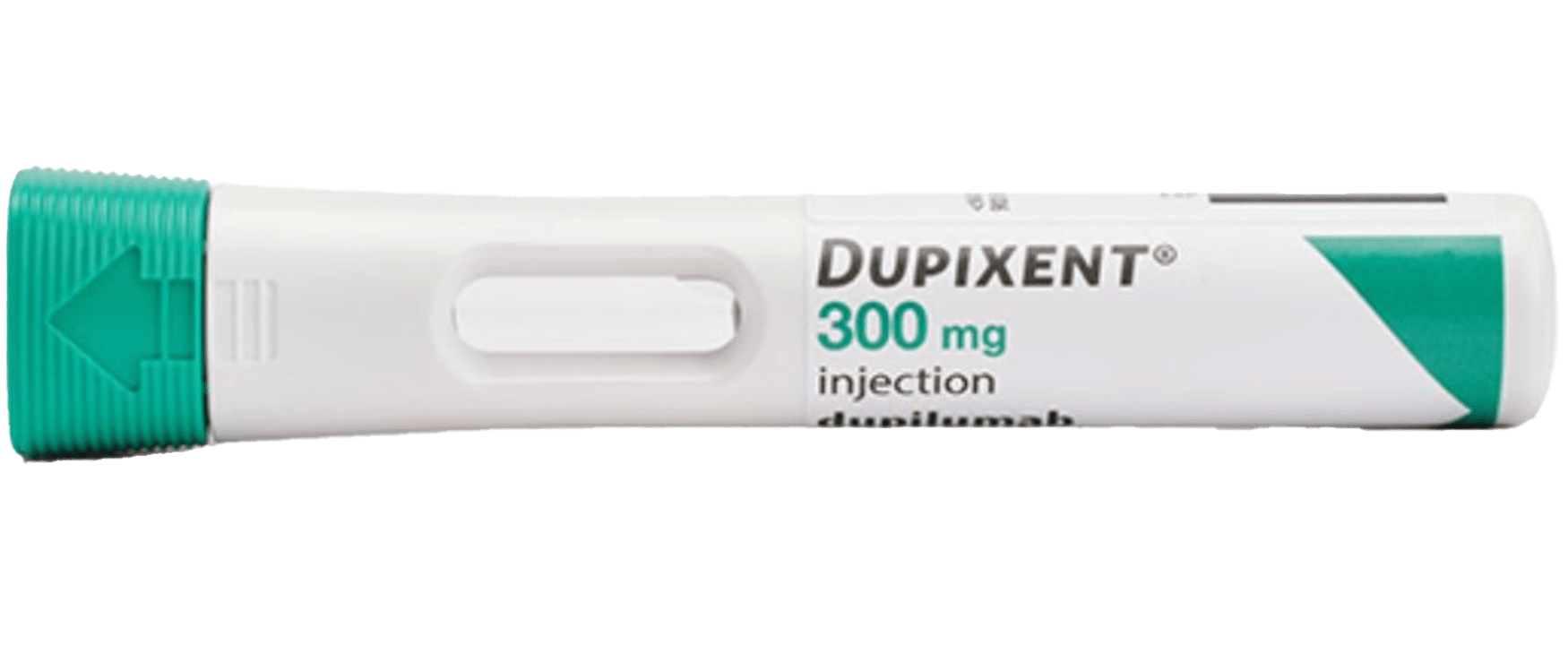
- Single-press auto-injector
- Visual and audible feedback
- Hidden needle
DUPIXENT Pre-filled Syringe2,3
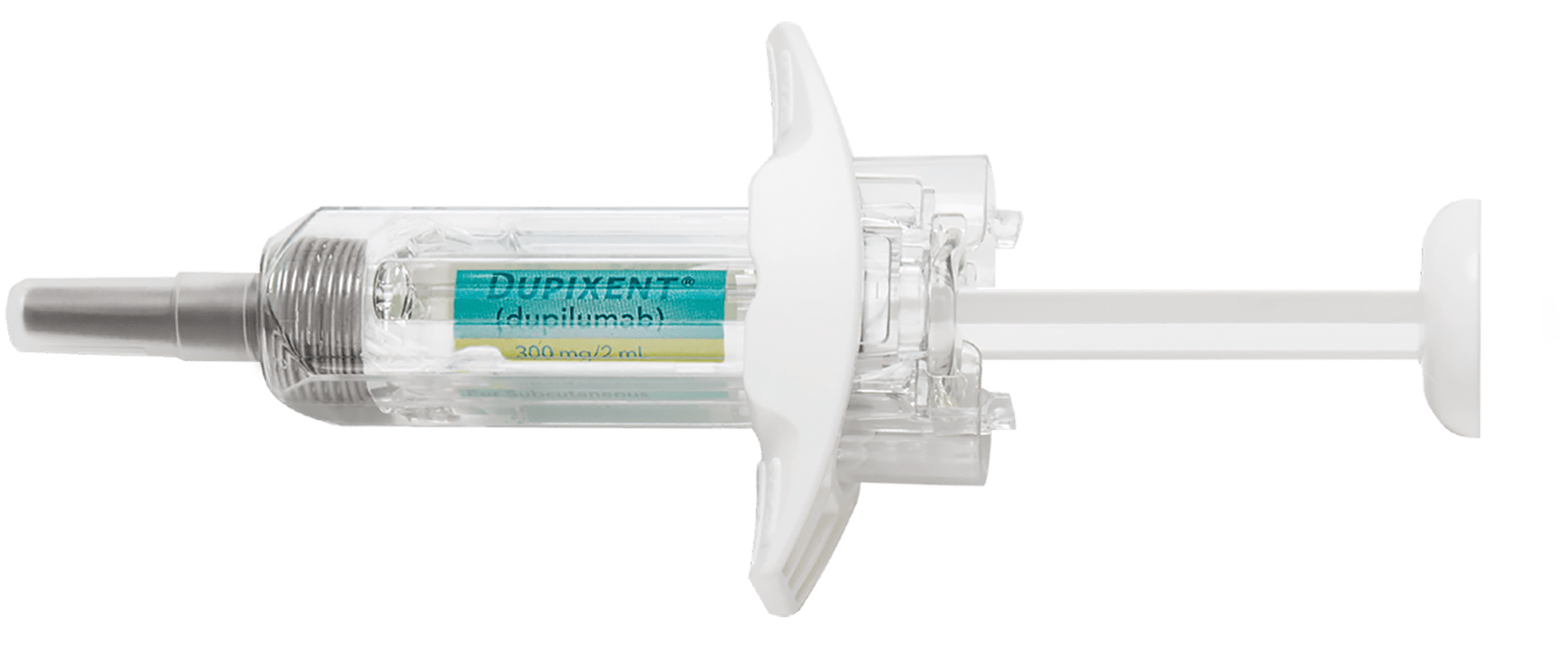
- Manual control of injection speed
- Finger grip for comfort
- Visual confirmation of injection delivery
- Needle shield
DUPIXENT is intended for use under the guidance of a healthcare provider1
- DUPIXENT can be injected by a patient or caregiver at home after training in a subcutaneous injection technique, using the auto-injection pen (for adults and adolescents) or pre-filled syringe if their healthcare professional determines this is appropriate.
- Physicians or nurses should provide proper training to patients and/or caregivers on the preparation and administration of DUPIXENT prior to use, according to the Instructions for Use
- DUPIXENT can be administered in the clinic under the guidance of a healthcare provider if the patient or caregiver is not an appropriate candidate to administer the injection
HOW TO INJECT DUPIXENT
DUPIXENT pre-filled pen injection
DUPIXENT pre-filled syringe injection
DUPIXENT offers a fixed dosing schedule with no loading dose1

a300 mg of dupilumab in 2 mL solution.1
Two devices to provide confident subcutaneous administration to your patients
DUPIXENT Pre-Filled Pen2,3

- Single-press auto-injector
- Visual and audible feedback
- Hidden needle
DUPIXENT Pre-filled Syringe2,3

- Manual control of injection speed
- Finger grip for comfort
- Visual confirmation of injection delivery
- Needle shield
DUPIXENT is intended for use under the guidance of a healthcare provider1
- DUPIXENT can be injected by a patient or caregiver at home after training in a subcutaneous injection technique, using the auto-injection pen (for adults and adolescents) or pre-filled syringe if their healthcare professional determines this is appropriate.
- Physicians or nurses should provide proper training to patients and/or caregivers on the preparation and administration of DUPIXENT prior to use, according to the Instructions for Use
- DUPIXENT can be administered in the clinic under the guidance of a healthcare provider if the patient or caregiver is not an appropriate candidate to administer the injection
HOW TO INJECT DUPIXENT
DUPIXENT pre-filled pen injection
DUPIXENT pre-filled syringe injection
For full administration instructions, please click here for the Summary of Product Characteristics.
Find resources and support for your eligible patients on DUPIXENT.
REFERENCES
- DUPIXENT (dupilumab). Summary of Product Characteristics. 2025. Date last accessed: January 2026.
- DUPIXENT (dupilumab). 300 mg solution for injection in pre-filled pen. Patient Information Leaflet. 2025. Date last accessed: January 2026.
- DUPIXENT (dupilumab). 300 mg solution for injection in pre-filled syringe. Patient Information Leaflet. 2025. Date last accessed: January 2026.

