Efficacy
MORE I CAN MOMENTS for eligible patients who continue to experience exacerbations on triple inhaled therapy, or double inhaled therapy if ICS is not appropriate.
A breakthrough therapy in COPD1
MORE I CAN MOMENTS with reduced exacerbations1
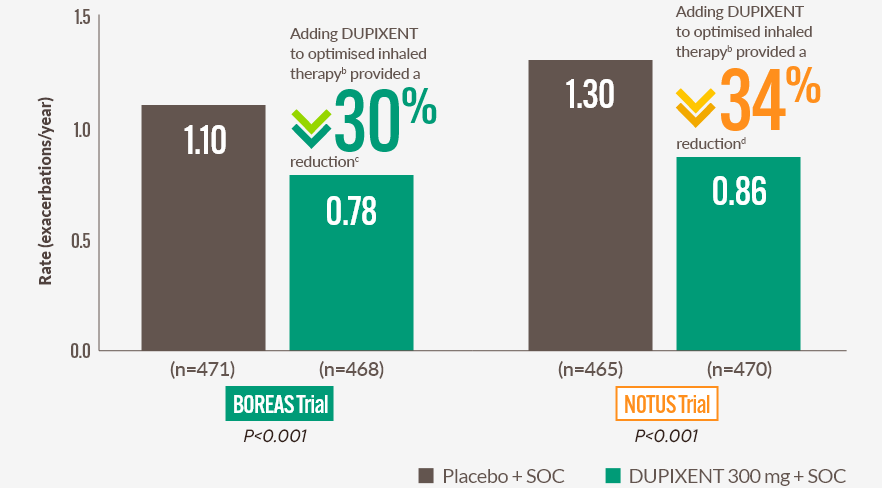
DUPIXENT + SOC consistently and significantly (P<0.001) reduced the annualised rate of moderate or severe COPD exacerbation rates in patients with uncontrolled COPD and type 2 inflammation vs placebo + SOCa,1
Moderate or severe exacerbations are a major contributor to poor outcomes in COPD2
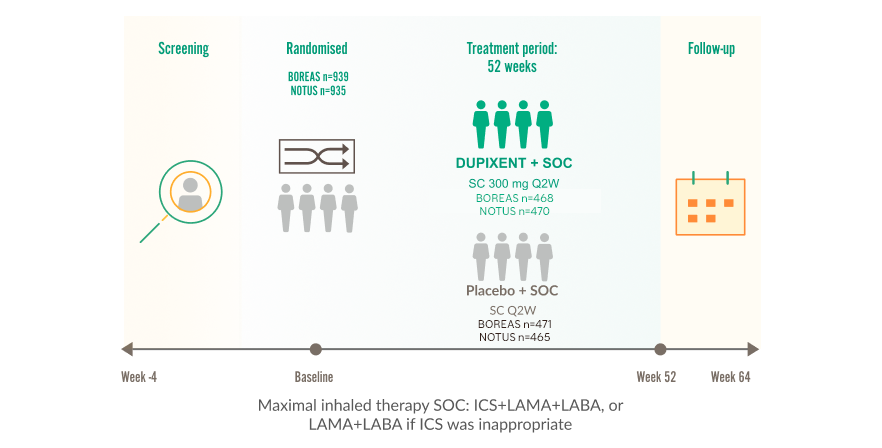
BOREAS and NOTUS clinical trials assessed safety, tolerability and efficacy in COPD patients with Type 2 Inflammation1,3,4
All patients received maximal inhaled therapy SOC (ICS+LAMA+LABA, or LAMA+LABA if ICS was not appropriate)l,1
Two phase 3, multicentre, international, double-blind, randomised, placebo-controlled trials in patients with uncontrolled COPD1,3,4
BOREAS and NOTUS Clinical Trial Designs
|
PRIMARY ENDPOINT |
Week 52 | Annualised rate of moderate or severe COPD exacerbations (primary endpoint was met) |
|
SELECT SECONDARY AND OTHER ENDPOINTS |
Week 12 Week 52 | Change in lung function (pre-BD FEV1) from baseline |
|
Week 52 | Change in SGRQ total score from baseline | |
|
Week 52 | Percentage of patients with SGRQ total score improvement ≥4 points | |
|
Week 12 Week 52 | Change in lung function (pre-BD FEV1) from baseline among patients with a baseline fractional exhaled nitric oxide (FeNO) ≥20 ppb | |
|
Week 52 | Annualised rate of moderate or severe exacerbations of COPD among patients with a baseline FeNO ≥20 ppb | |
|
Week 52 | Change in E-RS:COPD total score from baseline |
|
Key inclusion criteria3,4 |
| Physician-diagnosed COPD for ≥12 months prior to randomisation |
| Current or former smokers with a smoking history of ≥10 pack-years |
| Signs or symptoms of chronic bronchitis (chronic productive cough) for ≥3 months |
| ≥1 exacerbation while on SOC |
| Modified Medical Research Council dyspnoea scale grade ≥2 |
| Blood eosinophil counts ≥300 cells/μL |
| ≥2 moderate or ≥1 severe exacerbations within the year prior to screening |
| Background triple therapy (ICS+LAMA+LABA) for 3 months prior to randomisation with a stable dose of medication for ≥1 month prior to visit 1; double therapy (LAMA+LABA) allowed if ICS was not appropriate |
|
Key exclusion criteria3,4 |
| Physician-diagnosed COPD for <12 months prior to randomisation |
| Diagnosis or history of asthma according to the GINA guidelines |
'For more information, click here to view the DUPIXENT safety profile
|
Parameter |
BOREAS (n=939) |
NOTUS (n=935) |
| Mean age, years (±SD) | 65.1 (8.1) | 65.0 (8.3) |
| Male, % | 66.0 | 67.6 |
| White, %m | 84.1 | 89.6 |
| Mean smoking history, pack-years (±SD) | 40.5 (23.4) | 40.3 (27.2) |
| Current smokers, % | 30 | 29.5 |
| Emphysema, % | 32.6 | 30.4 |
| Mean duration of COPD, years (±SD) | 8.8 (6.0) | 9.3 (6.4) |
| Mean number of moderaten or severeo exacerbations in previous year (±SD) | 2.3 (1.0) | 2.1 (0.9) |
| Mean number of severe exacerbationso in previous year (±SD) | 0.3 (0.7) | 0.3 (0.6) |
| Background COPD medications at randomisation: ICS/LAMA/LABA (%) | 97.6 | 98.8 |
| Mean post-BD FEV1/FVC ratio (±SD) | 0.5 (0.1) | 0.5 (0.1) |
| Mean pre-BD FEV1 (L) (±SD) | 1.3 (0.5) | 1.4 (0.5) |
| Mean post-BD FEV1 (L) (±SD) | 1.4 (0.5) | 1.5 (0.5) |
| Mean percent predicted post-BD FEV1, % (±SD) | 50.6 (13.1) | 50.1 (12.6) |
| Mean percent predicted post-BD FEV1 <50%, % (±SD) | 467 (49.7) | 478 (51.3) |
| Mean SGRQ total score (±SD) | 48.4 (17.4) | 51.5 (17.0) |
| Mean E-RS:COPD, total score (±SD) | 12.9 (7.1) | 13.3 (7.0) |
| Mean BODE index score (± SD) | 4.1 (1.7) | 4.0 (1.6) |
| Mean FeNO, ppb (±SD) | 24.3 (22.4) | 24.6 (26.0) |
| Mean baseline blood eosinophil count, cells/μL (±SD) | 401 (298) | 407 (336) |
| Median baseline blood eosinophil count, cells/μL (interquartile range) | 340 (240–460) | 330 (220–460) |
l98% received ICS+LAMA+LABA in BOREAS and 99% in NOTUS.1
mIn BOREAS, 0.5% of participants were Black and 14.3% were Asian. In NOTUS, 1.3% of participants were Black and 1.1% were Asian.1
nModerate exacerbations were defined as exacerbations that resulted in treatment with systemic corticosteroids, antibiotics, or both.1
oSevere exacerbations were defined as exacerbations that resulted in hospitalisation or observation for over 24 hours in an emergency department or urgent care facility.1
|
Key inclusion criteria3,4 |
| Physician-diagnosed COPD for ≥12 months prior to randomisation |
| Current or former smokers with a smoking history of ≥10 pack-years |
| Signs or symptoms of chronic bronchitis (chronic productive cough) for ≥3 months |
| ≥1 exacerbation while on SOC |
| Modified Medical Research Council dyspnoea scale grade ≥2 |
| Blood eosinophil counts ≥300 cells/μL |
| ≥2 moderate or ≥1 severe exacerbations within the year prior to screening |
| Background triple therapy (ICS+LAMA+LABA) for 3 months prior to randomisation with a stable dose of medication for ≥1 month prior to visit 1; double therapy (LAMA+LABA) allowed if ICS was not appropriate |
|
Key exclusion criteria3,4 |
| Physician-diagnosed COPD for <12 months prior to randomisation |
| Diagnosis or history of asthma according to the GINA guidelines |
'For more information, click here to view the DUPIXENT safety profile
|
Parameter |
BOREAS (n=939) |
NOTUS (n=935) |
| Mean age, years (±SD) | 65.1 (8.1) | 65.0 (8.3) |
| Male, % | 66.0 | 67.6 |
| White, %m | 84.1 | 89.6 |
| Mean smoking history, pack-years (±SD) | 40.5 (23.4) | 40.3 (27.2) |
| Current smokers, % | 30 | 29.5 |
| Emphysema, % | 32.6 | 30.4 |
| Mean duration of COPD, years (±SD) | 8.8 (6.0) | 9.3 (6.4) |
| Mean number of moderaten or severeo exacerbations in previous year (±SD) | 2.3 (1.0) | 2.1 (0.9) |
| Mean number of severe exacerbationso in previous year (±SD) | 0.3 (0.7) | 0.3 (0.6) |
| Background COPD medications at randomisation: ICS/LAMA/LABA (%) | 97.6 | 98.8 |
| Mean post-BD FEV1/FVC ratio (±SD) | 0.5 (0.1) | 0.5 (0.1) |
| Mean pre-BD FEV1 (L) (±SD) | 1.3 (0.5) | 1.4 (0.5) |
| Mean post-BD FEV1 (L) (±SD) | 1.4 (0.5) | 1.5 (0.5) |
| Mean percent predicted post-BD FEV1, % (±SD) | 50.6 (13.1) | 50.1 (12.6) |
| Mean percent predicted post-BD FEV1 <50%, % (±SD) | 467 (49.7) | 478 (51.3) |
| Mean SGRQ total score (±SD) | 48.4 (17.4) | 51.5 (17.0) |
| Mean E-RS:COPD, total score (±SD) | 12.9 (7.1) | 13.3 (7.0) |
| Mean BODE index score (± SD) | 4.1 (1.7) | 4.0 (1.6) |
| Mean FeNO, ppb (±SD) | 24.3 (22.4) | 24.6 (26.0) |
| Mean baseline blood eosinophil count, cells/μL (±SD) | 401 (298) | 407 (336) |
| Median baseline blood eosinophil count, cells/μL (interquartile range) | 340 (240–460) | 330 (220–460) |
l98% received ICS+LAMA+LABA in BOREAS and 99% in NOTUS.1
mIn BOREAS, 0.5% of participants were Black and 14.3% were Asian. In NOTUS, 1.3% of participants were Black and 1.1% were Asian.1
nModerate exacerbations were defined as exacerbations that resulted in treatment with systemic corticosteroids, antibiotics, or both.1
oSevere exacerbations were defined as exacerbations that resulted in hospitalisation or observation for over 24 hours in an emergency department or urgent care facility.1
Annualised rate of moderate or severe COPD exacerbations at Week 52 (primary endpoint)1,3,4
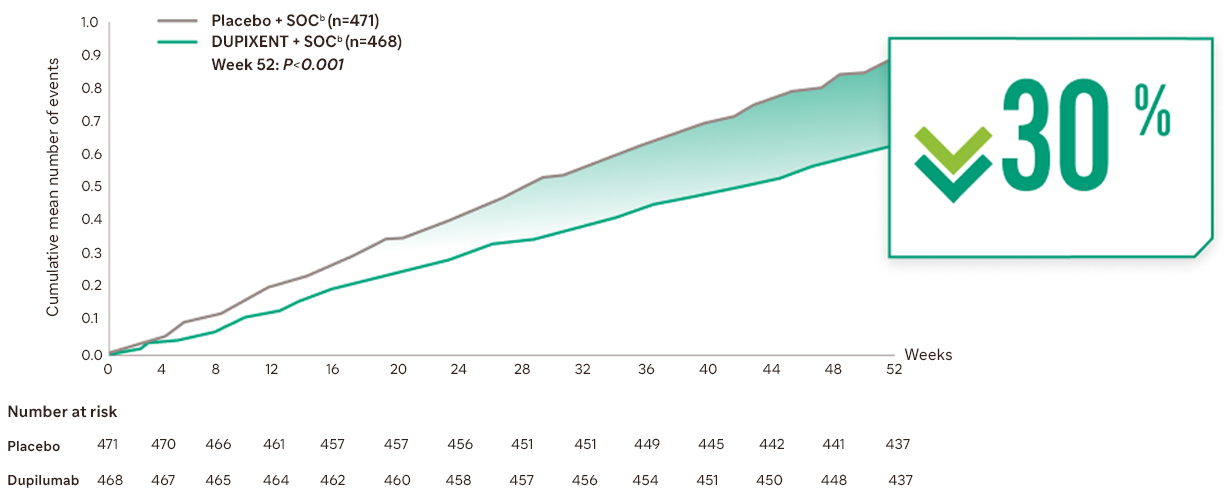
Cumulative mean number of moderate or severe COPD exacerbations over 52 weeksa,1
SOC, standard of care
Adding DUPIXENT to optimised inhaled therapy provided a reduction in moderate or severe COPD exacerbations vs placebo + SOC at Week 52a,b,1
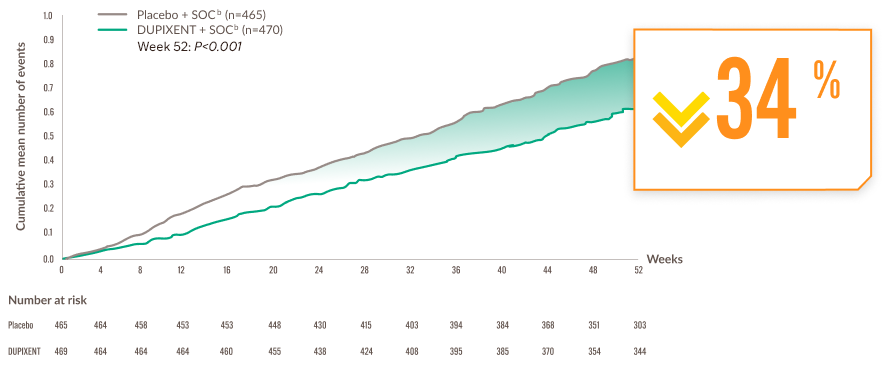
Cumulative mean number of moderate or severe COPD exacerbations over 52 weeksa,1
SOC, standard of care
Adding DUPIXENT to optimised inhaled therapy significantly reduced the annualised rate of moderate or severe COPD exacerbations vs placebo + SOC at Week 52a,b,1
Exacerbation reduction with DUPIXENT + SOC vs placebo + SOC seen across key patient subgroups regardless of:b,1,3,4

Number of moderate or severe exacerbationsa in prior year
(≤2, n=371 [BOREAS] and n=381 [NOTUS];
3, n=57 [BOREAS] and n=57 [NOTUS];
≥4, n=40 [BOREAS] and n=32 [NOTUS])e,3,4

Disease severity at baseline (post-BD FEV1 % predicted: <50%, n=227 [BOREAS] and n=250 [NOTUS] or ≥50%, n=241 [BOREAS] and n=217 [NOTUS])3,4

Smoking status at screening (former, n=334 [BOREAS] and n=328 [NOTUS]; current, n=134 [BOREAS] and n=142 [NOTUS])3,4
Cumulative mean number of moderate or severe COPD exacerbations over 52 weeksa,1

SOC, standard of care
Adding DUPIXENT to optimised inhaled therapy provided a reduction in moderate or severe COPD exacerbations vs placebo + SOC at Week 52a,b,1
Cumulative mean number of moderate or severe COPD exacerbations over 52 weeksa,1

SOC, standard of care
Adding DUPIXENT to optimised inhaled therapy significantly reduced the annualised rate of moderate or severe COPD exacerbations vs placebo + SOC at Week 52a,b,1
Exacerbation reduction with DUPIXENT + SOC vs placebo + SOC seen across key patient subgroups regardless of:b,1,3,4

Number of moderate or severe exacerbationsa in prior year
(≤2, n=371 [BOREAS] and n=381 [NOTUS];
3, n=57 [BOREAS] and n=57 [NOTUS];
≥4, n=40 [BOREAS] and n=32 [NOTUS])e,3,4

Disease severity at baseline (post-BD FEV1 % predicted: <50%, n=227 [BOREAS] and n=250 [NOTUS] or ≥50%, n=241 [BOREAS] and n=217 [NOTUS])3,4

Smoking status at screening (former, n=334 [BOREAS] and n=328 [NOTUS]; current, n=134 [BOREAS] and n=142 [NOTUS])3,4
aModerate exacerbations were defined as exacerbations that resulted in treatment with a systemic glucocorticoid, an antibiotic, or both. Severe exacerbations were defined as exacerbations that led to hospitalisation or an emergency medical care visit/urgent care visit with observation >24 h or that resulted in death.1,3-5
bSOC was optimised inhaled therapy; either triple inhaled therapy, or double inhaled therapy if ICS was not appropriate.1
cBOREAS (rate ratio vs placebo: 0.70, 95% CI 0.58 to 0.86).1
dNOTUS (rate ratio vs placebo: 0.66, 95% CI 0.54 to 0.82).1
eInclusion criteria required patients to have experienced ≥1 exacerbation while on SOC.3,4
MORE I CAN MOMENTS with improved breathing1
DUPIXENT + SOC provided sustained and clinically relevant improvementsf in lung functions vs placebo + SOC from baseline to Week 52
Mean change from baseline in pre-BD FEV1g,1
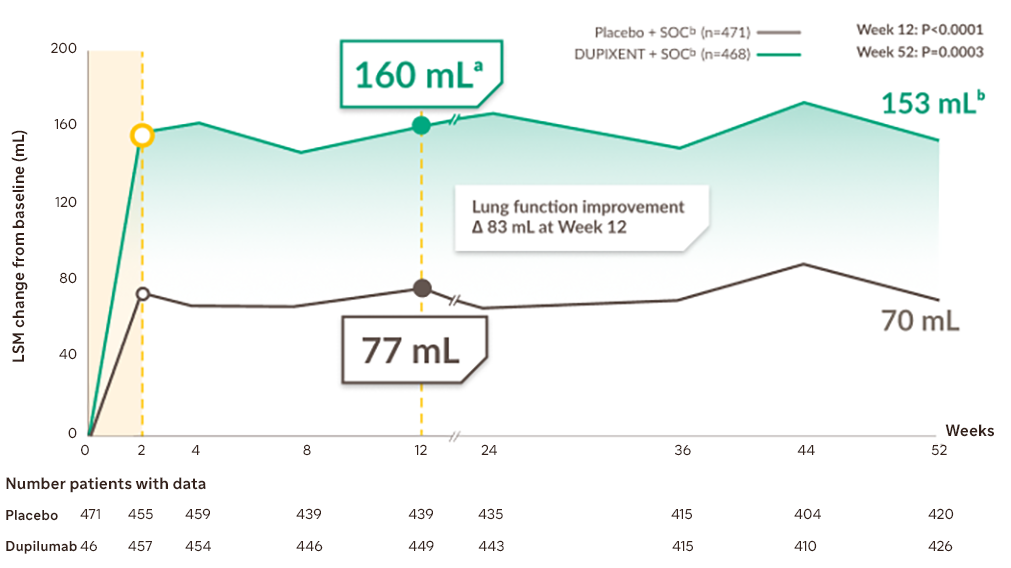
LSM, least squares mean; SOC, standard of care
bSOC was optimised inhaled therapy; either triple inhaled therapy, or double inhaled therapy if ICS was not appropriate.1
fFEV1 is a key spirometric measure used to assess lung function and treatment response in COPD. GOLD 2026 recognises spirometry as an accepted physiologic measure for COPD assessment and notes that interpretation of spirometric changes is based on established ERS/ATS technical standards. Recent ERS/ATS guidance has proposed thresholds for bronchodilator response, including a ≥10% change in predicted FEV1.6,7
gKey secondary endpoints were the change from baseline in pre-BD FEV1 at Week 12 and Week 52.1
Mean change from baseline in pre-BD FEV1g,1
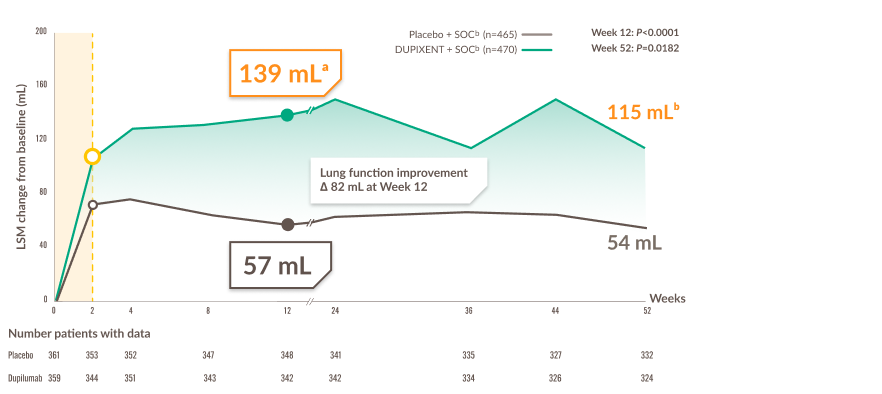
LSM, least squares mean; SOC, standard of care
bSOC was optimised inhaled therapy; either triple inhaled therapy, or double inhaled therapy if ICS was not appropriate.1
fFEV1 is a key spirometric measure used to assess lung function and treatment response in COPD. GOLD 2026 recognises spirometry as an accepted physiologic measure for COPD assessment and notes that interpretation of spirometric changes is based on established ERS/ATS technical standards. Recent ERS/ATS guidance has proposed thresholds for bronchodilator response, including a ≥10% change in predicted FEV1.6,7
gKey secondary endpoints were the change from baseline in pre-BD FEV1 at Week 12 and Week 52.1
Mean change from baseline in pre-BD FEV1g,1

LSM, least squares mean; SOC, standard of care
bSOC was optimised inhaled therapy; either triple inhaled therapy, or double inhaled therapy if ICS was not appropriate.1
fFEV1 is a key spirometric measure used to assess lung function and treatment response in COPD. GOLD 2026 recognises spirometry as an accepted physiologic measure for COPD assessment and notes that interpretation of spirometric changes is based on established ERS/ATS technical standards. Recent ERS/ATS guidance has proposed thresholds for bronchodilator response, including a ≥10% change in predicted FEV1.6,7
gKey secondary endpoints were the change from baseline in pre-BD FEV1 at Week 12 and Week 52.1
Mean change from baseline in pre-BD FEV1g,1

LSM, least squares mean; SOC, standard of care
bSOC was optimised inhaled therapy; either triple inhaled therapy, or double inhaled therapy if ICS was not appropriate.1
fFEV1 is a key spirometric measure used to assess lung function and treatment response in COPD. GOLD 2026 recognises spirometry as an accepted physiologic measure for COPD assessment and notes that interpretation of spirometric changes is based on established ERS/ATS technical standards. Recent ERS/ATS guidance has proposed thresholds for bronchodilator response, including a ≥10% change in predicted FEV1.6,7
gKey secondary endpoints were the change from baseline in pre-BD FEV1 at Week 12 and Week 52.1
Improved quality of life with DUPIXENT vs placebo for MORE I CAN MOMENTS1
DUPIXENT + SOC significantly improved quality of life for most patients with COPD vs placebo + SOCb,1,3,4
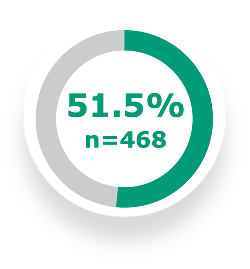
51.5% of patients on DUPIXENT in addition to triple inhaled therapy, or double inhaled therapy if ICS was not appropriate, achieved a clinically meaningful response in SGRQg total score (≥4 points) vs 43.1% with placebo + SOC (n=471) at Week 52 (P=0.009)3
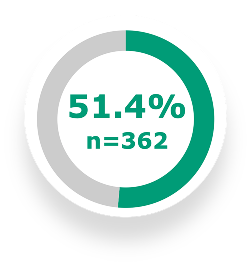
In the NOTUS trial, 51.4% of patients on update to DUPIXENT + SOC (n=362) achieved a nominally improved SGRQ total score vs 46.5% with placebo + SOC (n=359) at Week 52 (OR: 1.16; 95% CI: 0.86 to 1.58)4

51.5% of patients on DUPIXENT in addition to triple inhaled therapy, or double inhaled therapy if ICS was not appropriate, achieved a clinically meaningful response in SGRQg total score (≥4 points) vs 43.1% with placebo + SOC (n=471) at Week 52 (P=0.009)3

In the NOTUS trial, 51.4% of patients on update to DUPIXENT + SOC (n=362) achieved a nominally improved SGRQ total score vs 46.5% with placebo + SOC (n=359) at Week 52 (OR: 1.16; 95% CI: 0.86 to 1.58)4
Improvement in quality of life across 52 weeks of treatment1,3
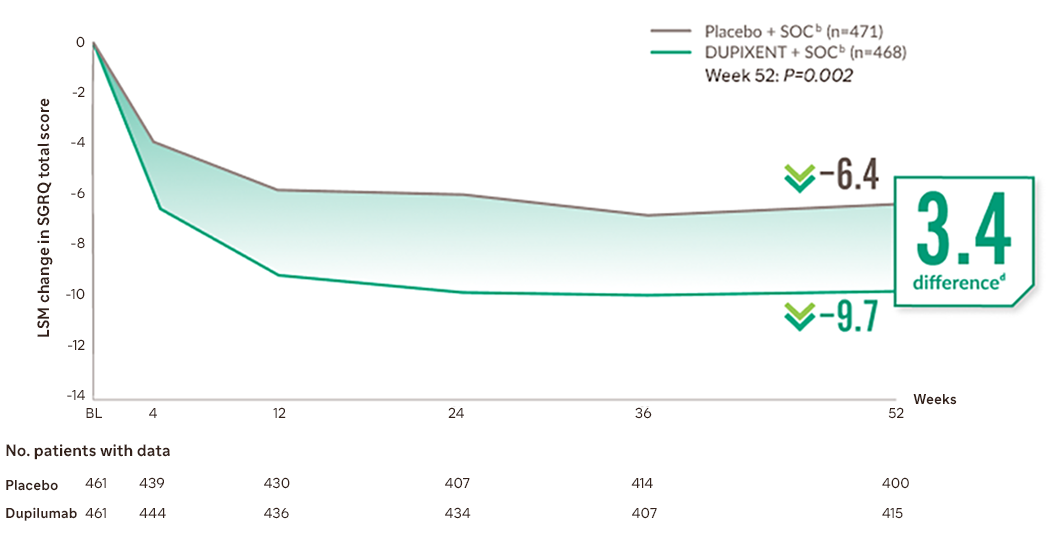
LSM, least squares mean; SGRQ, St George’s Respiratory Questionnaire; SOC, standard of care
At Week 52 in the NOTUS trial, patients on DUPIXENT + SOC experienced a nominal LSM difference in SGRQ total score compared with placebo + SOC of 3.37 points (95% CI: –5.81 to –0.93; P=0.007)1
bSOC was optimised inhaled therapy; either triple inhaled therapy, or double inhaled therapy if ICS was not appropriate.1
hThe SGRQ is a 50-item questionnaire designed to measure and quantify health status in adult patients with chronic airflow limitation. Scores range from 0 to 100, with lower scores indicating a better quality of life.8
iBOREAS, Week 52: LSM difference -3.36 (95% CI -5.46 to -1.27; P=0.0017).1
Select areas of SGRQ measurement8
Improve symptoms for MORE I CAN MOMENTS with DUPIXENT3
DUPIXENT + SOC improved symptoms that impact patient wellbeing vs placebo + SOCb,j,3
BOREAS Trial
Symptoms were assessed using E-RS:COPDj,1,3,4
E-RS:COPD, Evaluating Respiratory Symptoms in COPD.
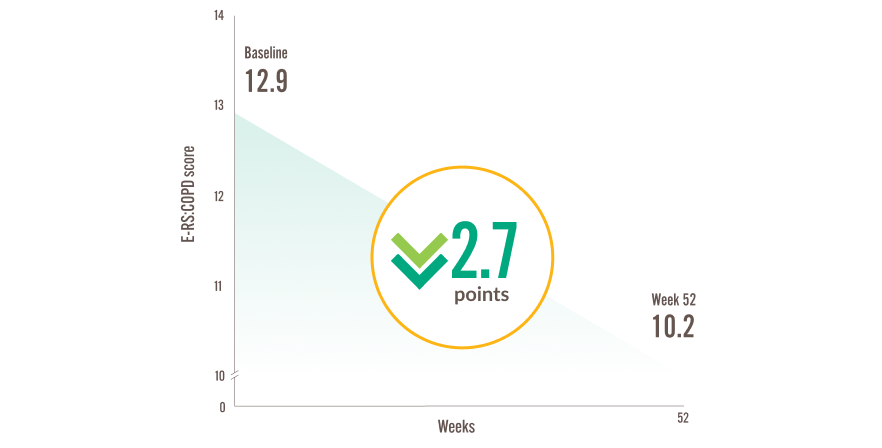
Mean change from baseline to Week 52 in E-RS:COPD total score (exploratory endpoint)b,3
E-RS:COPD, Evaluating Respiratory Symptoms in COPD
In the NOTUS trial, E-RS:COPD scores declined by 2.4 points (95% CI —3.0 to —1.8) with DUPIXENT + SOC (n=362) vs a 1.8-points (95% CI —2.4 to —1.2) decline with placebo + SOC (n=359) at Week 52b,4
In the BOREAS trial, E-RS:COPD scores declined by 2.7 points (95% CI —3.2 to —2.2) with DUPIXENT + SOC (n=468) vs a 1.6-point (95% CI, —2.1 to —1.1) decline with placebo + SOC (n=471) at Week 52 (P=0.001)b,3
The E-RS:COPD total clinically meaningful score change threshold is a ≥2.0-point reduction9–11
bSOC was triple inhaled therapy, or double inhaled therapy if ICS was not appropriate.1
jE-RS:COPD was an exploratory endpoint of the NOTUS and BOREAS trials.3 E-RS:COPD utilizes the 11 patient-reported respiratory symptom items contained in the 14-item Exacerbations of COPD Tool (EXACT). Scores range from 0-40, with a lower score indicating lower disease severity.9
ABBREVIATIONS
ATS, American Thoracic Society; BD, bronchodilator; BODE, body mass index, airflow obstruction, dyspnoea, and exercise capacity; CI, confidence interval; COPD, chronic obstructive pulmonary disease; ERS, European Respiratory Society; E‑RS:COPD, Evaluating Respiratory Symptoms in COPD; EXACT, EXAcerbations of COPD Tool; FeNO, fractional exhaled nitric oxide; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; GINA, Global Initiative for Asthma; ICS, inhaled corticosteroid; LABA, long‑acting beta2‑agonist; LAMA, long‑acting muscarinic antagonist; LSM, least squares mean; OR, odds ratio; Q2W, once every 2 weeks; SD, standard deviation; SGRQ, St George’s Respiratory Questionnaire; SOC, standard of care
REFERENCES
- DUPIXENT (dupilumab). Summary of Product Characteristics. 2025. Date last accessed: January 2026.
- Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease (2025 report). Available at: https://goldcopd.org/2025-gold-report/ Date last accessed: January 2026.
- Bhatt SP, et al. N Engl J Med. 2023;389(3):205‑214.
- Bhatt SP, et al. N Engl J Med. 2024;390(24):2274‑2283.
- Bhatt SP, et al. Lancet Respir Med. 2025;13:234‑243.
- GOLD. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease.
2026 report. Available at: https://goldcopd.org/2026-gold-report-and-pocket-guide/. - Stanojevic S, et al. Eur Respir J. 2022;60(1):2101499.
- St George’s University of London. St George’s Respiratory Questionnaire Manual. 2022. Available at: https://www.sgul.ac.uk/research/research-operations/research-administration/st-georges-respiratory-questionnaire/docs/SGRQ-Manual-March-2022.pdf Date last accessed: January 2026.
- Pharmaceutical Product Development. EXACT Program. Available at: https://www.evidera.com/what-we-do/patient-centered-research/coa-instrument-management-services/exact-program/exact-content/ Date last accessed: January 2026.
- Leidy NK, et al. Chronic Obstr Pulm Dis. 2022;9(4):576-590.
- Bushnell DM, et al. Chronic Obstr Pulm Dis. 2021;8(4):551‑571.

