- Article
- Source: Campus Sanofi
- 15 May 2025
Real-world evidence
The results from real world data analyses are limited by potential selection bias, as treatment groups often show differences in baseline characteristics. Outcome definitions may differ between RCTs and RWD analyses, and clinical events cannot be adjudicated. Certain clinical parameters may not be present in observational data; these analyses are subject to limitations.
Beyfortus® has a body of real-world evidence against RSV disease, including its impact on hospitalisations
>50 real-world studies in over 400,000 infants across the Northern and Southern Hemispheresa,1–59
Watch Prof. Saul N Faust and Dr Katrina Cathie explore the Spanish and Irish real-world evidence for Beyfortus®

Professor Saul N Faust
Professor of Paediatric Immunology & Infectious Diseases; NIHR Southampton Clinical Research Facility and Biomedical Research Centre, University Hospital Southampton, NHS Foundation Trust; Faculty of Medicine and Institute for Life Sciences, University of Southampton, Southampton.

Dr Katrina Cathie
Consultant Paediatrician; Associate Director, NIHR Southampton Clinical Research Facility; Southampton Children's Hospital Speciality Group Clinical Lead for Paediatrics in Wessex, Chair of the UK Children's General Paediatrics Clinical Studies Group.
NIRSE-GAL, Spain1
Nirse-GAL study (Galicia, Spain) measured the effectiveness in preventing hospitalisation and public health impact of Beyfortus® in infants born during and before RSV season1a
High coverage rates were achieved with Beyfortus® in a universal prophylaxis strategy for eligible infants in Galicia2
Population-based longitudinal cohort study (N=14,476 eligible infants)c

Primary endpoint: Beyfortus® effectiveness against RSV-LRTI hospitalisationb,2
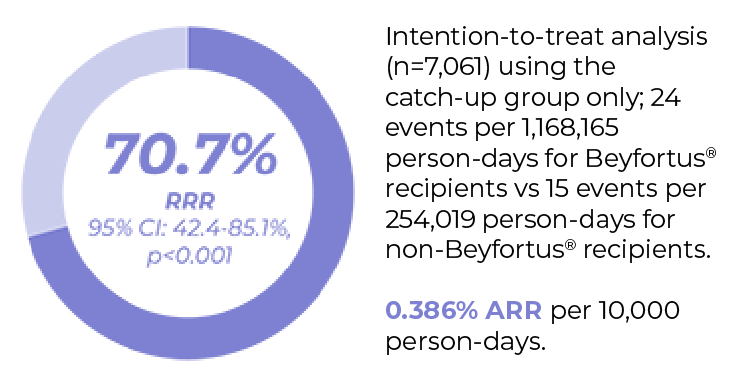
No serious adverse events directly linked to Beyfortus® were reportedd, consistent with clinical trial results1,60,61
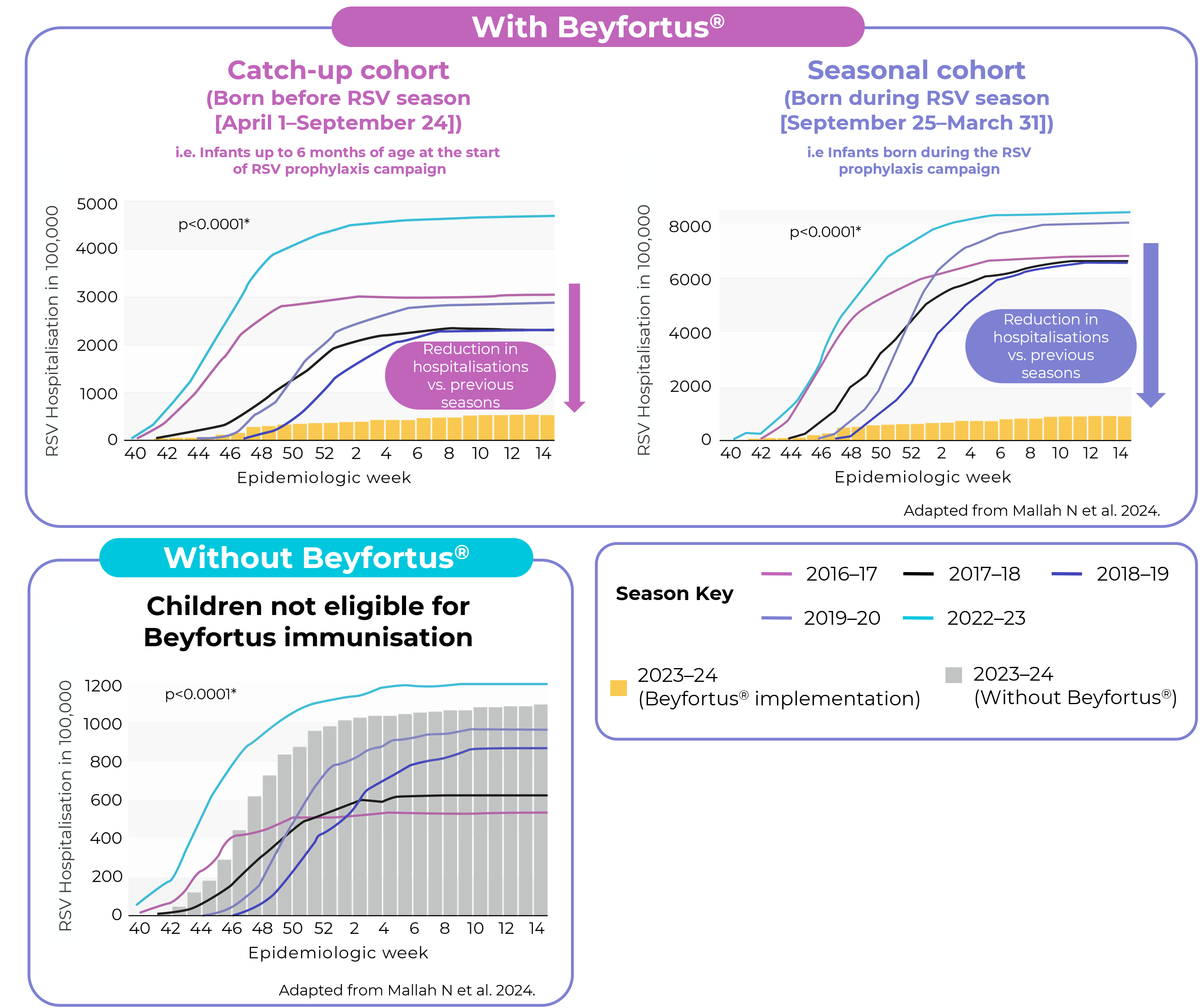
Public health impact of Beyfortus® on RSV hospitalisation in Galicia, Spain
After Beyfortus® implementation median RSV LRTI hospitalisation decreased by 89.2% (RRR; IQR: 89.1% - 91.4%; ARR: 0.386)e vs. five previous seasons for infants in the overall cohort (infants born before and during RSV season)b2
EPI-PHARE, France62
Objective: To compare the associations of maternal vaccination with the RSVpreFf vaccine vs passive infant immunisation with Beyfortus® for the prevention of RSV-related hospitalisation.
In this study, Beyfortus® was associated with a lower risk of RSV hospitalisation and severe outcomes compared to RSVpreF.*

*These findings reflect the first RSV season with use of these immunisation strategies in mainland France; their use should be re-evaluated in future studies.
**Inverse probability of treatment weighting based on the propensity score was applied to reduce confounding and improve between-group covariate comparability. Weighted conditional Cox proportional hazards models were fitted to ac- count for the matched design and estimate hazard ratios(HRs) with 95% CIs.
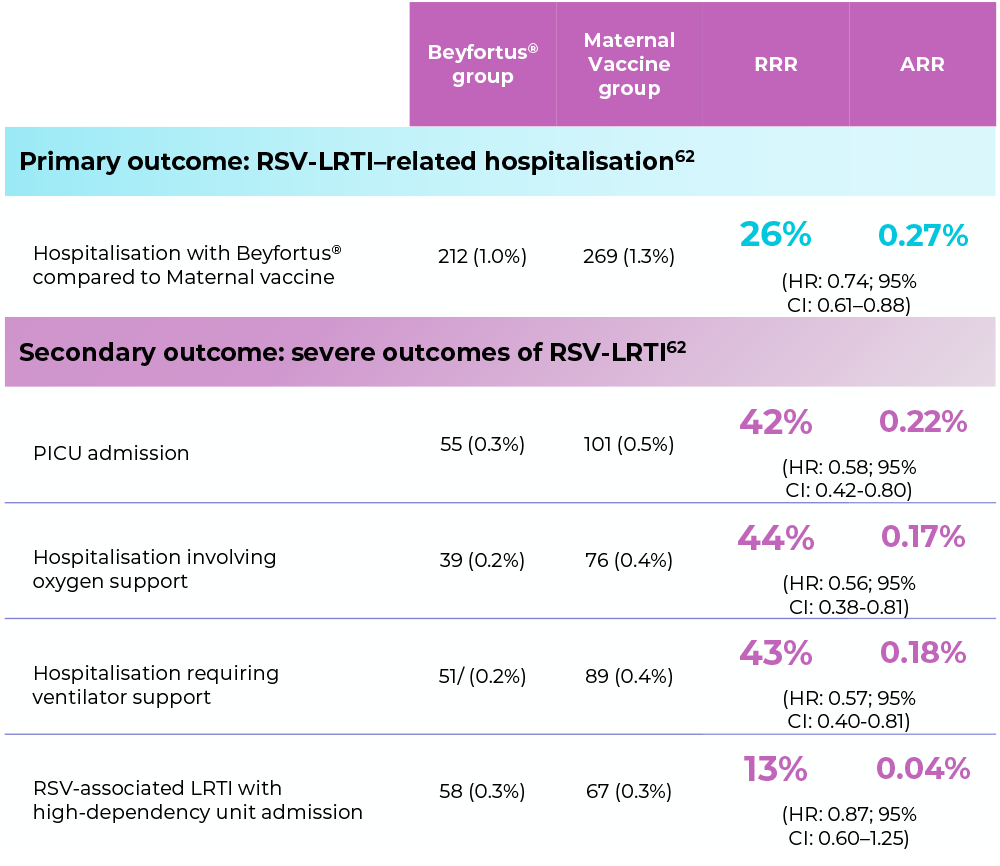
Total infants under each group: 21,280. This study did not assess the safety of Beyfortus® or maternal vaccine.62 In controlled clinical trials, both Beyfortus® and maternal vaccine have demonstrated efficacy in terms of RSV-LRTD prevention with acceptable safety profiles.63-64
Severe outcomes included the need for PICU admission, ventilator support, and oxygen therapy.
Strengths62
- French National Health Data System was used, offering comprehensive, longitudinal healthcare data for the entire population
- Included detailed maternal and infant characteristics, enabling extensive adjustment for potential confounders
- Aligned the timing of eligibility assessment, treatment assignment, and follow-up within the same RSV epidemic period and population
- Provided an opportunity to compare maternal vaccination and infant Beyfortus® immunisation under real-world conditions, addressing a critical evidence gap as these strategies are implemented internationally
Limitations62
- Infants were born later in the RSV season due to vaccine availability and matching by birth date, which may limit generalisability to those born earlier
- Conducted during the first season of maternal vaccination in France, with eligibility restricted to 32–36 weeks’ gestation, unlike the WHO’s recommendation of starting at 28 weeks. Findings reflect early national experience and require confirmation in future studies and settings
- Subgroup analyses were only stratified up to 60 days, limiting insights into longer-term differences
- Potential unmeasured confounding factors (e.g, parental health- seeking behavior, household smoking, and day care attendance) could not be excluded
Protecting infants from RSV in Ireland: Impact of a national immunisation programme65
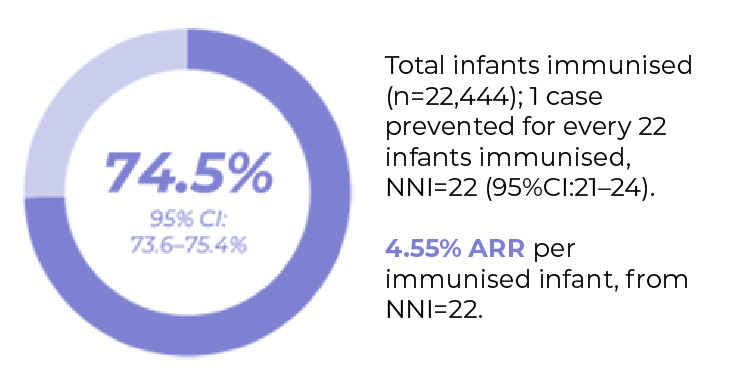
This study shows the impact of a national Beyfortus® immunisation programme in Ireland for infants born during the 2024/2025 RSV seasong.
Retrospective, population-based longitudinal ecological study in infants born during the RSV programme period. (seasonal) (n=22,444 immunised/26,903 eligible infants).
Aim of the study: Impact of Beyfortus® immunisation programme on RSV related morbidity
High coverage rate was achieved with Beyfortus® in Ireland

In comparison to the previous two years, seasonal immunisation with Beyfortus® in 2024/2025 helped to avoid:
- 1,055 RSV cases (95% CI:1038-1071)
- 437 Hospitalisations (95% CI:430-443)
- 76 ICU admissions (95% CI:74-78)
A reduction in RSV-related outcomes was demonstrated by disease prevented fraction with Beyfortus®

AE, adverse event; ARR, absolute risk reduction; CI, confidence interval; GA, gestational age; HR, hazard ratio; LRTI, lower respiratory tract disease; RRR, relative risk reduction; RSV, respiratory syncytial virus; PICU, paediatric intensive care unit; RCT, randomised controlled trial; RWD, real-world data; NNI, number needed to immunise.
a. In Andorra, Australia, Chile, France, Italy, Luxembourg, Portugal, Spain, and the United States as of August 05, 2025.
b. Effectiveness and impact were estimated using the seasonal and catch-up groups. Nirsevimab effectiveness was estimated from incidence rate ratios calculated using Poisson regression models, which were adjusted for enrolment group (catch-up and seasonal), sex, and health district area. Only patients with non-zero follow-up time were included.1
c. Immunisation campaign: September 25, 2023-March 31, 2024. Eligible infants in Galicia included those aged <6 months at the start of the immunisation campaign (born between April 1 and September 24, 2023 [catch-up group]) and those born during the immunisation campaign (born between September 25 2023 and March 31 2024 [seasonal group]. A high-risk group was also immunised (n=348/360) as part of the Galician immunisation campaign. This group was not included in effectiveness or public health impact calculations.1
d. Adverse events related to Beyfortus® administration were routinely monitored through the Galician pharmacovigilance system. In addition, active surveillance of any potential adverse event or hospitalisation in the first 3 weeks after Beyfortus administration was conducted in the pre-term population (infants with a GA <37 weeks).1
e. P-values were obtained from linear regression model analysis of weekly accumulated incidence rates comparing the 2023-2024.
f. Abrysvo (respiratory syncytial virus vaccine [bivalent, recombinant]), Pfizer.63
g. The target population for impact estimation was infants born between the immunisation programme, and vulnerable infants who were eligible to receive nirsevimab through Child's Health Ireland. High-risk infants (born prior to 1st September 2024) that received Beyfortus® in homecare settings (n=399, 1.5%) were not included in this study. To estimate immunisation impact, an effectiveness estimate of 88.4% was used from a published systematic review and meta-analysis66 and total cumulative national immunisation uptake rates to estimate averted RSV-related outcomes applying adapted Machado et al. formulas.67 This included averted cases for outcomes of interest, disease prevented fraction (PF), and the number needed to immunise (NNI) to prevent one RSV case, ED presentation, hospitalisation, and ICU admission.
- Ares-Gómez S et al. Lancet Infect Dis 2024; 24(8): 817–828.
- Mallah N et al. Lancet Infect Dis 2024; 25(2): e62–e63 & Supplementary Appendix.
- Ernst C et al. Euro Surveill 2024; 29(4): 2400033.
- Ezpeleta G et al. Vaccines 2024; 12(4): 383.
- López-Lacort M et al. Euro Surveill 2024; 29(6): 2400046.
- Paireau J et al. Influenza Other Respir Viruses 2024; 18(6): e13311.
- Mazagatos C et al. Influenza Other Resp Viruses 2024; 18(5): e13294.
- Consolati A et al. Vaccines 2024; 12(5): 549.
- Estrella-Porter P et al. Vaccines 2024; 42(22): 126030.
- Levy C et al. J Pediatr Infect Dis Soc 2024; 13(7): 371–373.
- Coma E et al. Arch Dis Child 2024; 109(9): 736–741.
- Agüera M et al. Pediatr Allergy Immunol 2024; 35(6): e14175.
- Assad Z et al. N Engl J Med 2024; 391(2): 144–154.
- Molina Gutierrez MA et al. Enferm Infecc Microbiol Clin (Engl Ed) 2024; 42(7): 367–372.
- Lassoued Y et al. Lancet Reg Health Eur 2024; 44: 101007.
- Barbas Del Buey JF et al. Front Public Health 2024; 12: 1441786.
- Pérez Martín JJ and Moreno MZ. Human Vaccines Immunother 2024; 20(1): 2365804.
- García-García ML et al Authorea 2024. https://doi.org/10.22541/au.172464773.33348406/v1. [Epub ahead of print].
- Carbajal R et al. Lancet Child Adolesc Health 2024; 8: 730–739.
- Martinez DA et al. Pediatrics 2024; 154(4): e2024066584.
- Xu H et al. JAMA Netw Open 2025; 8(3): e250380.
- Jimeno Ruiz S et al. Vaccines 2024; 12: 1160.
- López-Lacort M et al. Pediatrics 2025; 155(1): e2024066393.
- Perramon-Malavez A et al. Euro J Pediatr 2024; 183: 5181–5189.
- Reina J. Enferm Infecc Microbiol Clin 2023; 41: 301–304.
- Torres JP et al. Universal Immunization Strategy Against Respiratory Syncytial Virus (RSV) Prevention in Chile with Nirsevimab during the 2024 Winter Season: First Southern Hemisphere Nationwide Effectiveness Data. Open Forum Infectious diseases; 12(Supplement 1): February 2025. Available at: https://academic.oup.com/ofid/article/12/Supplement_1/ofae631.006/7986730. Accessed: March 2026.
- Hsiao A et al. Annals Allergy Asthma Immunol 2024; 133(6;Supple2): S3–S4.
- Lefferts B et al. Morbidity and Mortality Weekly Report 2024; 73(45): 1015–1021.
- Jeziorski E et al. Pediatr Infect Dis J 2024; 44(4): e124–e126.
- Askari MS et al. Morbidity and Mortality Weekly Report 2024; 73(48): 1107–1109.
- De Sentuary CO et al. EClinicalMedicine 2024; 79: 102986.
- Moline HL et al. JAMA Pediatr 2025; 179-187.
- Chauvel C et al. Influenza Other Respi Viruses 2024; 18: e70054.
- Ma KSK et al. Pediatr Res 2025. doi: 10.1038/s41390-024-03782-4. [Epub ahead of print].
- Carcione D et al. Pediatr Infect Dis J 2025. doi: 10.1097/INF.0000000000004715. [Epub ahead of print].
- Perramon-Malavez A et al. Pediatr Infect Dis J 2025. doi: 10.1097/INF.0000000000004672. [Epub ahead of print].
- Cavaco Afonso AF et al. Authorea 2025. doi: 10.22541/au.173780129.99727424/v1. [Epub ahead of print].
- Lenglart L et al. Eur J Pediatr 2025; 184(2):171.
- Núñez O et al. Euro Surveill 2025; 30(5): 2400596.
- Litman A et al. JAMA Netw Open 2025; 8(2): e2460729.
- Jabagi MJ et al. NEJM Evid 2025; 4(3): doi: 10.1056/EVIDoa2400275.
- Moreno-Pérez D et al. Vaccines 2025; 13(2): 175.
- Pastor-Barriuso R et al. Euro Surveill 2025; 30(6): 2500040.
- Blauvelt CA et al. JAMA Net Op 2025; 8(2): e2460735.
- Marouk, A et al. Eur J Pediatr 184, 229 (2025).
- Wadia U et al. J Infect. 2025 Apr;90(4):106466.
- Rius-Peris JM, et al. Acta Paediatr. 2025 Aug;114(8):1963-1976.
- Touati, S et al. Ann. Intensive Care 15, 56 (2025).
- Bermúdez-Barrezueta, L et al. Eur J Pediatr 184, 345 (2025).
- Irving SA et al. Pediatrics. 2025 Jun 1;155(6):e2024070240.
- Silva-Afonso RF et al. Enferm Infecc Microbiol Clin (Engl Ed). 2025 Jun 3:S2529-993X(25)00139-X.
- Coma E et al. Arch Dis Child. 2025 Jun 10: archdischild-2025-329005.
- Alejandre, C et al. Eur J Pediatr 183, 3897–3904 (2024).
- Cantais A, et al. J Med Virol. 2024 Feb;96(2):e29483.
- Espeleta-Fox A, et al. Pediatr Pulmonol. 2024 Dec;59(12):3783-3786.
- Homo RL, et al. J Pediatr. 2024 Oct;273:114144.
- Kemp M, et al. Vaccine. 2025 Feb 15;47:126674.
- Puckett L, et al. Hosp Pediatr. 2025 Feb 1;15(2):99-107.
- Razzaghi H, et al MMWR Morb Mortal Wkly Rep. 2024 Sep 26;73(38):837-843.
- Griffin MP et al. N Engl J Med 2020; 383(5): 415–425.
- Hammitt LL et al. N Engl J Med 2022; 386(9): 837–846.
- Jabagi MJ et al. JAMA. Published online December 22, 2025. doi:10.1001/jama.2025.24082.
- Abrysvo Summary of Product Characteristics (SmPC).
- Beyfortus® Summary of Product Characteristics (SmPC).
- Paris L. et al. Protecting infants from respiratory syncytial virus (RSV) in Ireland: Impact of a national nirsevimab immunisation programme, 2024/2025. Available at SSRN: https://ssrn.com/abstract=5445717 or http://dx.doi.org/10.2139/ssrn.5445717.
- Riccò M. et al. Impact of Nirsevimab Immunization on Pediatric Hospitalization Rates: A Systematic Review and Meta-Analysis. Vaccines (Basel). 2024 Jun 8;12(6):640.
- Machado, A. et al. Impact of national influenza vaccination strategy in severe influenza outcomes among the high-risk Portuguese population. BMC Public Health, 19(1), p.1690.
MAT-XU-2501637 (v3.0) Date of preparation: April 2026
