Breaking the von Willebrand Factor–Imposed Half-Life Ceiling
The half-life of most FVIII replacement therapies is limited by endogenous vWF binding, which imposes a half-life ceiling of ~15 hours.2
ALTUVIIIO is a first-in-class high-sustained FVIII replacement therapy designed to overcome half-life limitations, in part by decoupling rFVIII from endogenous vWF.1,3
ALTUVIIIO is a novel fusion protein composed of a single recombinant FVIII protein fused to the Fc domain of IgG1, a D'D3 domain of vWF, and 2 XTEN polypeptide insertions.1,2
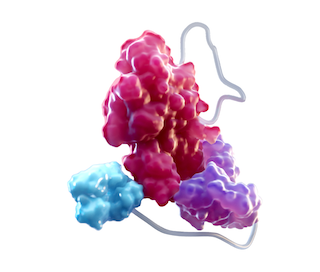
RECOMBINANT FACTOR VIII2,4
rFVIII is at the core of the ALTUVIIIO molecular structure*
- rFVIII replaces FVIII in hemophilia A patients to restore hemostasis
The half-life of ALTUVIIIO is increased through 3 additional components:
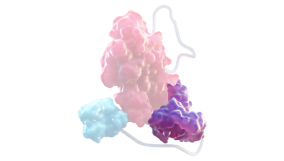
vWF D’D3 DOMAIN2,5
- Prevents rFVIII from interacting with endogenous vWF, to overcome the half-life ceiling
- Retains stabilization and protective functions of endogenous vWF
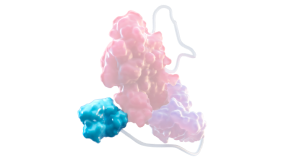
Fc DOMAIN1,2,6
- Enables recycling via the neonatal Fc receptor, which reduces lysosomal degradation of rFVIII
- Slows rFVIII clearance to extend its time in circulation
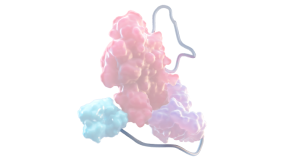
XTEN POLYPEPTIDE INSERTION1,2,6
- Increases the hydrodynamic size of the molecule to limit renal and receptor-mediated clearance
- Provides steric shielding to rFVIII, reducing proteolytic degradation
* ALTUVIIIO is composed of naturally occurring amino acids that are able to be broken down through regular protein degradation processes.4
Discover how ALTUVIIIO achieves higher sustained mean Factor VIII levels (>40%)1-3
In a Phase 3 study, ALTUVIIIO offered a 48.2-hour half life in adults—the longest of any Factor VIII replacement therapy.2,3,7-16†
† Comparison of data from XTEND-1 with half-life data recorded in prescribing information of other approved Factor VIII replacement therapies.
Indication
Fc=fragment crystallizable; IgG1=immunoglobulin G1; rFVIII=recombinant Factor VIII; vWF=von Willebrand Factor.
References: 1. von Drygalski A, et al. N Engl J Med. 2023;388(4):310-318. 2. Konkle BA, et al. N Engl J Med. 2020;383(11):1018-1027. 3. Wong N, et al. J Clin Pharmacol. 2025;65(7):860-872. 4. ALTUVIIIO Prescribing Information. Bioverativ Therapeutics Inc. Waltham, MA. 5. Chhabra ES, et al. Blood. 2020;135(17):1484-1496. 6. von Drygalski A, et al. Supplementary Appendix Protocol. N Engl J Med. 2023;388(4):310-318. 7. Adynovate Prescribing Information. Takeda Pharmaceuticals U.S.A., Inc. Lexington, MA. 8. Afstyla Prescribing Information. CSL Behring LLC. Kankakee, IL. 9. Eloctate Prescribing Information. Bioverativ Therapeutics Inc. Waltham, MA. 10. Esperoct Prescribing Information. Novo Nordisk Inc. Plainsboro, NJ. 11. Jivi Prescribing Information. Bayer HealthCare LLC. Whippany, NJ. 12. Kovaltry Prescribing Information. Bayer HealthCare LLC. Whippany, NJ. 13. Novoeight Prescribing Information. Novo Nordisk Inc. Plainsboro, NJ. 14. Nuwiq Prescribing Information. Octapharma USA, Inc. Paramus, NJ. 15. Recombinate Prescribing Information. Takeda Pharmaceuticals U.S.A., Inc. Lexington, MA. 16. Xyntha Prescribing Information. Wyeth Pharmaceuticals LLC. Philadelphia, PA.