- Article
- Source: Campus Sanofi
- 17 Sep 2025
Burden of Disease
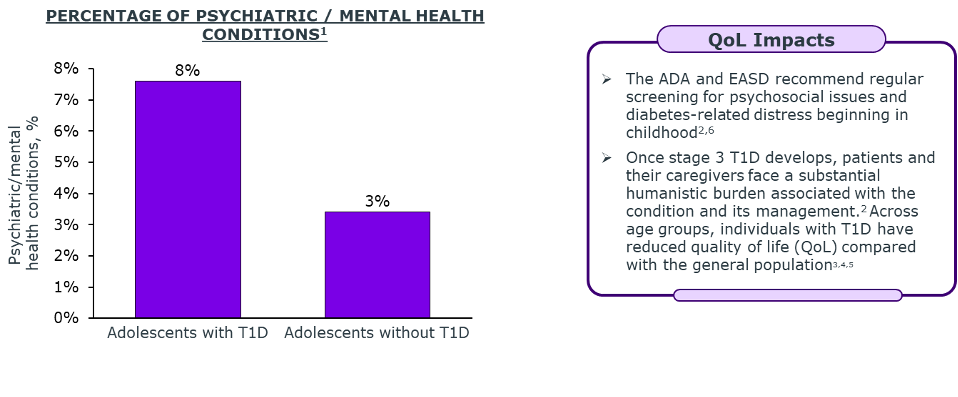
Onset of stage 3 aT1D at a younger age is associated with increased CVD risk and early mortality.4
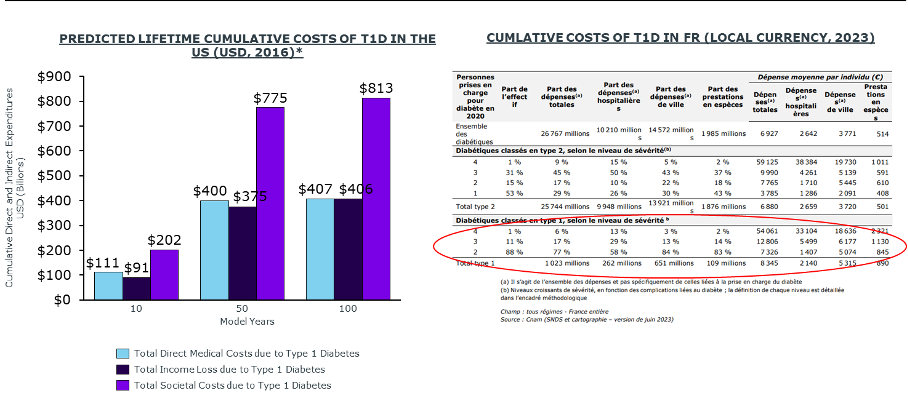
Lifelong management of aT1D drives significant increases in both direct (HCRU) and indirect costs (productivity loss, caregiver burden).5
- Besser REJ, Bell KJ, Couper JJ, et al. ISPAD Clinical Practice Consensus Guidelines 2022: Stages of type 1 diabetes in children and adolescents. Pediatr Diabetes. Dec 2022;23(8):1175-1187. doi:10.1111/pedi.13410; 3: Sparring V, Nyström L, Wahlström R, Jonsson PM, Ostman J, Burström K. Diabetes duration and health-related quality of life in individuals with onset of diabetes in the age group 15-34 years - a Swedish population-based study using EQ-5D. BMC Public Health. Apr 22 2013;13:377. doi:10.1186/1471-2458-13-377;
- Bronner MB, Peeters MAC, Sattoe JNT, van Staa A. The impact of type 1 diabetes on young adults' health-related quality of life. Health Qual Life Outcomes. May 12 2020;18(1):137. doi:10.1186/s12955-020-01370-8;
- Rechenberg K, Koerner Ms R. General Anxiety, Diabetes-Specific Anxiety, and Quality of Life in Adolescents With Type 1 Diabetes. Sci Diabetes Self Manag Care. Dec 2022;48(6):469-475. doi:10.1177/26350106221125689; 6: Holt RIG, DeVries JH, Hess-Fischl A, et al. The Management of Type 1 Diabetes in Adults. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. Nov 2021;44(11):2589-2625. doi:10.2337/dci21-0043
- Rawshani A, Sattar N, Franzén S, et al. Excess mortality and cardiovascular disease in young adults with type 1 diabetes in relation to age at onset: a nationwide, register-based cohort study. Lancet. Aug 11 2018;392(10146):477-486. doi:10.1016/s0140-6736(18)31506-x
- Sussman et al. 2020
GULF:
One JLT Building, Level 3, Jumeirah Lake Towers, PO Box 53899, Dubai, UAE .
For further medical information, please contact: For UAE ✆ 800 MEDICAL Toll-Free Number.
For all Gulf countries ✆ +971 45 50 38 63 or email: medical-information.gulf@sanofi.com.
Full prescribing information is available upon request.
To Report adverse events please call: +971 561747001 or email Gulf.Pharmacovigilance@sanofi.com
www.sanofi.com
KSA:
To report any side effect(s):
Saudi Arabia: The National Pharmacovigilance and Drug Safety Centre (NPC
SFDA call center: 19999 • E-mail: npc.drug@sfda.gov.sa • Website: https://ade.sfda.gov.sa/
Full Prescribing Information is available upon request: SANOFI, Kingdom of Saudi Arabia, P.O. Box 9874, Jeddah 21423, K.S.A.
Tel: +966-12-669-3318, Fax: +966-12-663-6191
For Medical Information, please contact: +966-12-669-3318, ksa.medicalinformation@sanofi.com
For Pharmacovigilance, please contact: +966-54-428-4797, ksa_pharmacovigilance@sanofi.com
To report any Product Technical Complaint, please contact SANOFI Quality Department:
Email: quality.greatergulf@Sanofi.com , www.sanofi.com.sa
MAT-KW-2600001-V1-Feb 2026