- Article
- Source: Campus Sanofi
- 18 Sep 2025
Tzield MOA and Dosing
Tzield is the first and only disease modifying immunotherapy developed for aT1D that treats the underlying cause of the disease by preserving beta-cell function.1-3
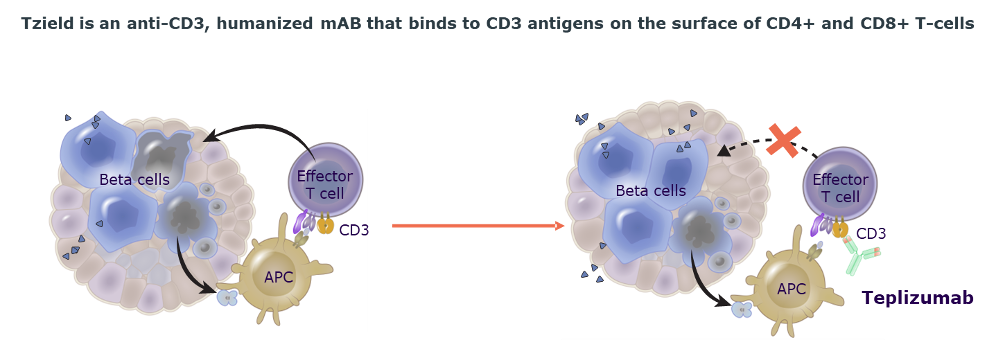
Unlike other chronic diabetes therapies, Tzield is a finite course of treatment. For Stage 2,
Tzield is administered through a single cycle of 14-day IV infusion.5
- Teplizumab is administered by IV (over a minimum of 30 minutes) once daily for 14 consecutive days
- The recommended dose for adults and pediatric patients aged ≥8 years is based on BSA according to the following:
- Two doses should not be administered on the same day
- If a planned teplizumab infusion is missed, resume dosing by administering all remaining doses on consecutive days to complete the 14-day treatment course

Unlike other chronic diabetes therapies, Tzield is a finite course of treatment. For Stage 2,
Tzield is administered through a single cycle of 14-day IV infusion.5
- Teplizumab is administered by IV (over a minimum of 30 minutes) once daily for 14 consecutive days
- The recommended dose for adults and pediatric patients aged ≥8 years is based on BSA according to the following:
- Two doses should not be administered on the same day
- If a planned teplizumab infusion is missed, resume dosing by administering all remaining doses on consecutive days to complete the 14-day treatment course
- Kuhn and Weiner, 2016;
- Chen and Flies, 2013.
- Murphy K, Weaver C. Janeway’s Immunobiology. 9th ed. Garland Science; 2017:257-93. Kuhn C, Weiner HL. Immunotherapy. 2016;8(8):889-906; 3. Long A, et al. Sci Immunol, 2016;1:eaai7793.
- Herold KC, et al. N Engl J Med 2019;381:603–13.
- TZIELD. Prescribing Information. Provention Bio, Inc
GULF:
One JLT Building, Level 3, Jumeirah Lake Towers, PO Box 53899, Dubai, UAE .
For further medical information, please contact: For UAE ✆ 800 MEDICAL Toll-Free Number.
For all Gulf countries ✆ +971 45 50 38 63 or email: medical-information.gulf@sanofi.com.
Full prescribing information is available upon request.
To Report adverse events please call: +971 561747001 or email Gulf.Pharmacovigilance@sanofi.com
www.sanofi.com
KSA:
To report any side effect(s):
Saudi Arabia: The National Pharmacovigilance and Drug Safety Centre (NPC
SFDA call center: 19999 • E-mail: npc.drug@sfda.gov.sa • Website: https://ade.sfda.gov.sa/
Full Prescribing Information is available upon request: SANOFI, Kingdom of Saudi Arabia, P.O. Box 9874, Jeddah 21423, K.S.A.
Tel: +966-12-669-3318, Fax: +966-12-663-6191
For Medical Information, please contact: +966-12-669-3318, ksa.medicalinformation@sanofi.com
For Pharmacovigilance, please contact: +966-54-428-4797, ksa_pharmacovigilance@sanofi.com
To report any Product Technical Complaint, please contact SANOFI Quality Department:
Email: quality.greatergulf@Sanofi.com , www.sanofi.com.sa
MAT-KW-2600001-V1-Feb 2026