Study Design
Superiority in long-lasting osteoarthritis (OA) knee pain relief demonstrated by head-to-head clinical trials against saline control and other treatments1-4
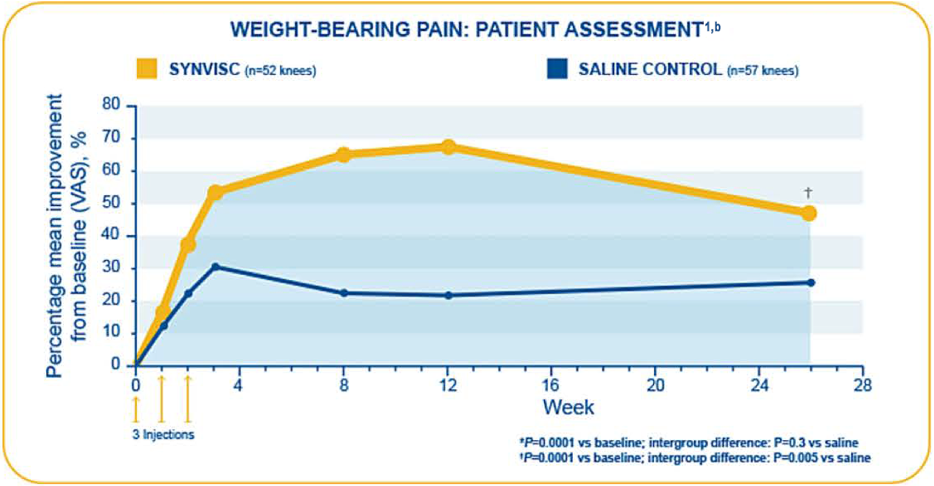
Pivotal study: Wobig et al.1,2,a
- This randomized, double-blind, clinical trial assessed the efficacy and safety of three 2-mL injections of SYNVISC given at 1-week intervals in patients with chronic OA of the knee
- Primary endpoints (assessed by patients as measured by VAS [0-100 mm]): Pain during weight-bearing, pain at rest during the night
- In this trial, SYNVISC patients had an average reduction of weight-bearing pain of 48.8% from baseline at 6 months, compared to 25% for saline-treated patients (P=0.005)
- Week 26 mean improvements from baseline for:
- Weight-bearing pain evaluated by patients: 34 mm (SYNVISC) and 19.1 mm (saline) (P=0.005)
- Pain at rest during night evaluated by patients: 24.3 mm (SYNVISC) and 12.8 mm (saline) (P=0.03)
- There were no reports of local adverse reactions to SYNVISC in the injected knee. In clinical trials, the most commonly reported adverse events (AEs) were transient pain, swelling, and or joint effusion in the injected knee. The following reported AEs are among those that may occur in association with intra-articular injections, including SYNVISC: arthralgia, joint stiffness, joint effusion, joint swelling, joint warmth, injection site pain, arthritis, arthropathy, and gait disturbance
aN=109 (3 weekly, 2-mL injections SYNVISC [n=52] or saline [n=57]). The most commonly reported device-related AEs were transient pain, swelling, and joint effusion in the injected knee.
.png)
bN=109 (3 weekly 2-mL injections SYNVISC [n=52] or saline [n=57]). Week 26 mean improvement from baseline for weight-bearing pain evaluated by patients: 34 mm (SYNVISC) and 19.1 mm (saline). Week 26 data based on patient telephone interviews.
Adverse Events Involving the Injected Joint1
Clinical Trials
A total of 511 patients (559 knees) received 1771 injections in seven clinical trials of SYNVISC®. There were 39 reports in 37 patients (2.2% of injections, 7.2% of patients) of knee pain and/or swelling after these injections. Ten patients (10 knees) were treated with arthrocentesis and removal of joint effusion. Two additional patients (two knees) received treatment with intra-articular steroids. Two patients (two knees) received NSAIDs. One of these patients also received arthrocentesis. One patient was treated with arthroscopy. The remaining patients with adverse events localized to the knee received no treatment or only analgesics. A total of 157 patients have received 553 injections in the three clinical trials of repeated courses of SYNVISC® treatment. The reports in these trials describe a total of 48 reports of adverse events localized to the injected knee in 35 patients that occurred after injections that patients had received during their second course of treatment. These adverse events accounted for 6.3% of injections in 22.3% of patients as compared to 2.2% of injections in 7.2% of patients in a single course of SYNVISC® injections.
Most commonly reported device-related AEs with SYNVISC1:
- Pain in the injected knee
- Swelling in the injected knee
- Joint effusion
Potential AEs that may occur in association with intra-articular injection, including SYNVISC1:
- Arthralgia
- Joint stiffness
- Joint effusion
- Joint swelling
- Joint warmth
- Injection site pain
- Arthritis
- Arthropathy
- Gait disturbance
Additional Data
SYNVISC® (hylan G-F 20) patient satisfaction
Based on a large, observational study to assess its tolerability over 80% of patients were satisfied, rating it as a “good” or “very good” therapy at 3 weeks after the first injection of SYNVISC5
of patients were satisfied with SYNVISC as their therapy at 3 weeks after the first injection5
TREATMENT-RELATED AEs5
- Treatment-related AEs were reported in 4.2% of patients (2.4% of injections)
- Most frequently reported local treatment-related AEs were joint effusion 91 (2.14%), joint swelling 48 (1.13%), arthralgia 42 (0.99%), joint warmth 23 (0.54%) and injection site erythema 13 (0.31%).
STUDY DESIGN5
- This prospective, observational study evaluated the tolerability of SYNVISC in the treatment of OA knee pain
- The primary objective was to report the tolerability of SYNVISC in German OA knee patients by documenting the incidence and type of local
- As an observational study, routine treatment of the patient for OA knee pain was not changed, and the protocol did not specify any additional diagnostic measures over and above those of routine practice
- 4253 patients received a total of 12,699 intra-articular injections of SYNVISC given by 840 orthopedic surgeons at 720 sites in Germany
- Tolerability was assessed by the occurrence of AEs, both local and systemic, including severity and relation to treatment, at visits 2, 3, and 4 (3 weeks after the first injection)
- Patients assessed their pain on a 4-point scale (none, mild, moderate, severe) prior to and 3 weeks after the first injection. At visit 4, patients rated a change in their pain (much better, better, no change, worse, much worse) with therapy, and both patients and physicians provided a general assessment of SYNVISC therapy
STUDY LIMITATIONS5
- Study lacked control group, which could potentially introduce both patient and investigator bias. Because the study focus was on tolerability and patients, the effectiveness of SYNVISC could only be interpreted on a short-term basis. Assessment of local AE occurrence with previous HA or SYNVISC use was limited due to brand of HA used unknown timing of prior treatment in relation to current therapy. OA diagnosis criteria were not recorded.
Study endpoints and measures differ from the pivotal trials. No conclusions of statistical or clinical significance can be drawn.
Important Safety Information
References
-
SYNVISC [prescribing information]. Cambridge, MA: Genzyme Corporation.
-
Wobig M, Dickhut A, Maier R, Vetter G. Viscosupplementation with Hylan G-F 20: a 26-week controlled trial of efficacy and safety in the osteoarthritic knee. Clin Ther. 1998;20(3):410-423.
-
Raman R, Dutta A, Day N, Sharma HK, Shaw CJ, Johnson GV. Efficacy of hylan G-F 20 and sodium hyaluronate in the treatment of osteoarthritis of the knee—a prospective randomized clinical trial. Knee. 2008;15(4):318-324.
-
Caborn D, Rush J, Lanzer W, Parenti D, Murray C. A randomized, single-blind comparison of the efficacy and tolerability of hylan G-F 20 and triamcinolone hexacetonide in patients with osteoarthritis of the knee. J Rheumatol. 2004;31(2):333-343.
-
Kemper F, Gebhardt U, Meng T. Tolerability and short-term effectiveness of hylan G-F 20 in 4253 patients with osteoarthritis of the knee in clinical practice. Curr Med Res Opin. 2005;21(8):1261-1269.
