Study Design
Pivotal study: Chevalier et al.1,2,a
- This randomized, double-blind, placebo-controlled clinical trial assessed the efficacy and safety of one 6-mL injection of SYNVISC-ONE compared to 6 mL of phosphate-buffered saline in patients with chronic osteoarthritis (OA) of the knee
- Primary endpoint: Change from baseline over 26 weeks in WOMAC A (pain) score using 5-point Likert scale where 0=no pain and 4=extreme pain
- In this trial, SYNVISC-ONE patients had a statistically significant change in WOMAC A pain score over 6 months compared with saline-treated patients (36% versus 29%, respectively, P=0.047; ITT population)
- Week 26 mean improvements from baseline for WOMAC A pain scores: -0.84 (SYNVISC-ONE) and -0.69 (saline)
- The incidence of Adverse events (AEs) was similar in the 2 groups (SYNVISC-ONE, n=70, 56.9%; saline, n=79, 60.8%).The most commonly reported AEs were arthralgia, arthritis, arthropathy, injection site pain, and joint effusion. The following reported adverse events are among those that may occur in association with intra-articular injections, including SYNVISC-ONE: arthralgia, joint stiffness, joint effusion, joint swelling, joint warmth, injection site pain, arthritis, arthropathy, and gait disturbance
aN=253 (6 mL of SYNVISC-ONE + arthrocentesis=124; 6 mL of phosphate-buffered saline placebo + arthrocentesis=129). The most commonly reported device-related AEs were pain in the target knee (coded as “arthralgia”), joint stiffness, joint effusion, arthritis, arthropathy, injection site pain, and joint swelling.
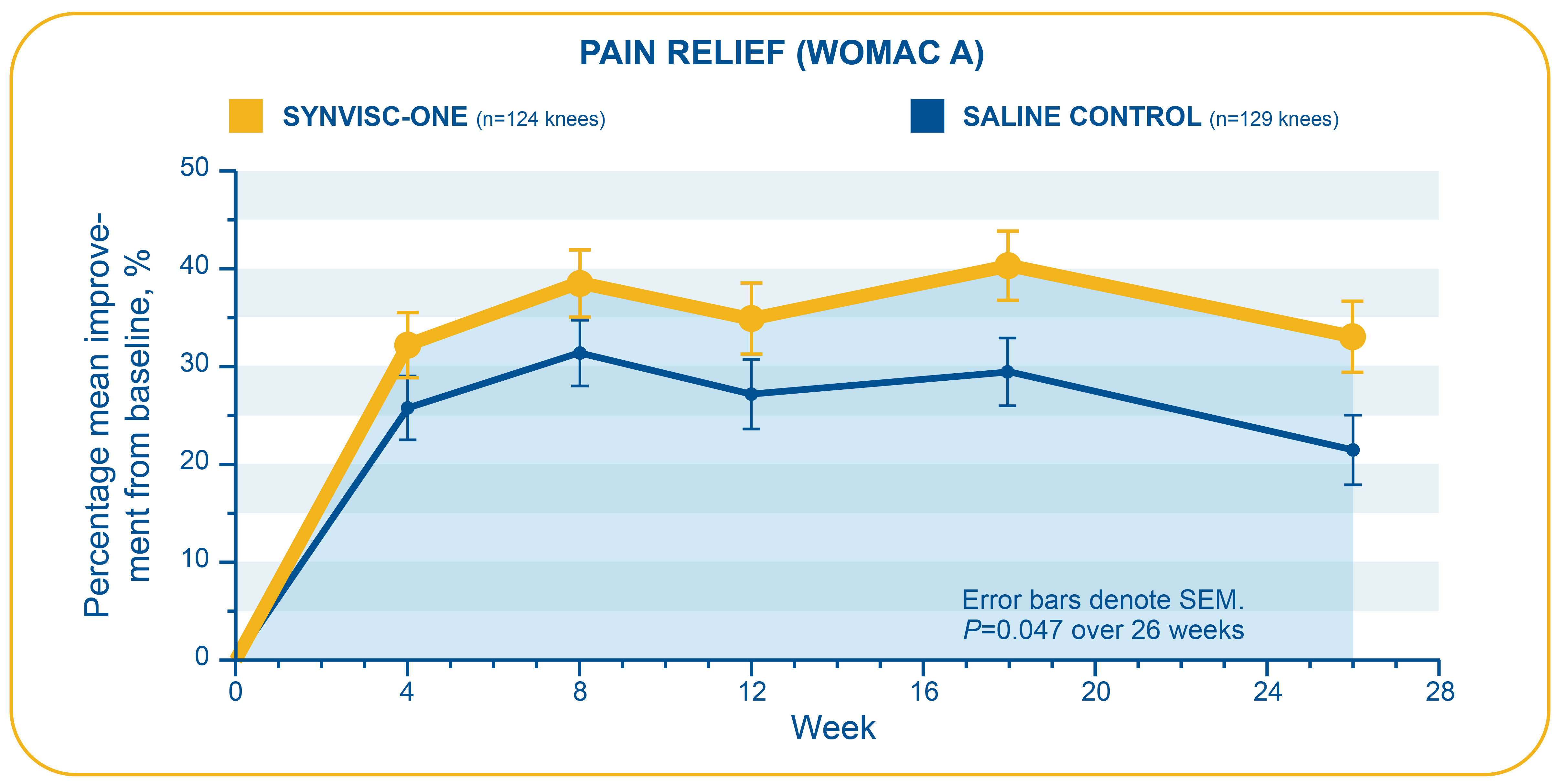
WOMAC=Western Ontario & McMaster Universities Osteoarthritis Index. WOMAC is an instrument used to assess changes in OA symptoms, with each question graded on a 5-point scale (0=low, 4=high).
Safety
The primary objective was to compare a single, 6 ml, intra-articular injection of hylan G-F 20 with placebo in patients with symptomatic knee OA. The safety of a repeat injection of hylan G-F 20 was also assessed.2
AEs and safety2
- There were no target knee serious AEs and no serious AEs that were related to the study treatment or the study procedure. The overall frequency of AEs was comparable between the two treatment groups (hylan G-F 20, n = 70, 56.9%; placebo, n = 79, 60.8%).
- The most commonly reported AEs were pain in the target knee (coded as “arthralgia”), joint stiffness, joint effusion and joint swelling. The incidence of AEs was slightly higher in the hylan G-F 20 group (n = 7, 5.7%) than in the placebo group (n = 4, 3.1%) but this was not statistically significant (p = 0.366) (table 1). In addition, there were no statistically significant differences between the groups in treatment-related (p = 0.203) or procedure-related (p = 0.531) target knee AEs, all of which were of mild or moderate severity. Pain, swelling and effusion were the most frequently occurring AEs in the injected knee. Cases of acute inflammation, characterized by joint pain, swelling, effusion and sometimes joint warmth and/or stiffness, have been reported following an intra-articular injection of SYNVISC-ONE®.
|
Table 1. Target knee adverse events: safety population | ||
| Preferred term |
Hylan G-F 20 N = 123 n (% of patients) |
Placebo N = 130 n (% of patients) |
| Any treatment-emergent target knee AEs | 44 (35.8) | 44 (33.8) |
| Any treatment and/or procedure-related target knee AEs | 7 (5.7) | 4 (3.1) |
| Arthralgia | 2 (1.6) | 3 (2.3) |
| Joint effusion | 2 (1.6) | 0 (0) |
| Arthritis | 1* (0.8) | 0 (0) |
| Arthropathy | 1 (0.8) | 0 (0) |
| Injection site pain | 1 (0.8) | 1 (0.8) |
| Any treatment-related target knee AEs | 4 (3.3) | 1 (0.8) |
| Any procedure-related target knee AEs | 6 (4.9) | 4 (3.1) |
| Related to treatment refers to unknown relationship to, or possibly, probably, or definitely related to treatment. Patients are counted once for each unique AEs and may have had more than one unique AEs. If a patient had more than one occurrence of the same AEs, the strongest relationship to study treatment or injection procedure was included. Treatment groups reflect the actual treatment received, not the randomised treatment. Patients may be counted in more than one category. *Patient withdrew from the study due to target knee arthritis of moderate severity. | ||
Repeat treatment phase2
- A total of 160 patients was treated in the open, repeat treatment phase, of which 77 received a second injection of hylan G-F 20 and 83 received a first injection of hylan G-F 20, having received placebo during the initial treatment phase. There were no target knee serious AEs. In the group receiving a second injection of hylan G-F 20 one patient (1.3%) experienced target knee AEs related to the study treatment and four patients (5.2%) experienced target knee AEs related to the study procedure.
- Patients who developed target knee AEs during the initial phase of the study, and who subsequently received repeat treatment, did not experience target knee AEs on repeat exposure to hylan G-F 20. All treatment-related and procedure-related target knee AEs were of mild or moderate severity.
Important Safety Information
References
-
Synvisc-One [prescribing information]. Cambridge, MA: Genzyme Corporation.
-
Chevalier X, Jerosch J, Goupille P, et al. Single, intra-articular treatment with 6 ml hylan G-F 20 in patients with symptomatic primary osteoarthritis of the knee: a randomised, multicentre, double-blind, placebo controlled trial. Ann Rheum Dis. 2010;69(1):113-119.
