How to Prepare Thymoglobulin® [Anti-thymocyte Globulin (Rabbit)] for Administration1
![Image of Thymoglobulin® [Anti‑thymocyte Globulin (Rabbit)] 25 mg vial next to its product carton.](https://pro.campus.sanofi/dam/jcr:8df4afa0-3250-4bfc-8d2a-b444a710a8f5/SAN-THYMO-img-%20calculate-dose-mobile.png)
Calculate dose based on body weight and determine the number of vials needed

Bring the appropriate number of vials to room temperature
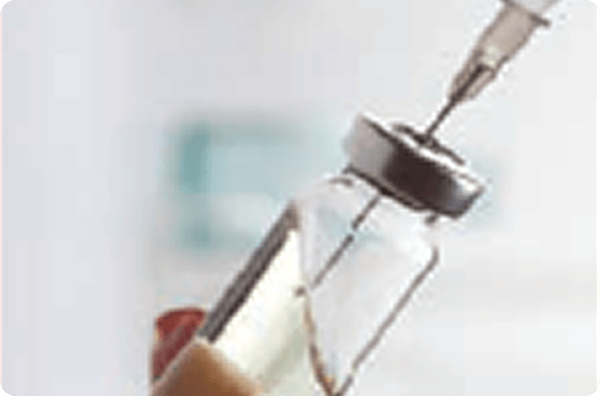
Reconstitute each vial with 5 ml of sterile water for injection using aseptic technique

Gently rotate vials until all product is dissolved. Inspect solution for particulate matter after reconstitution
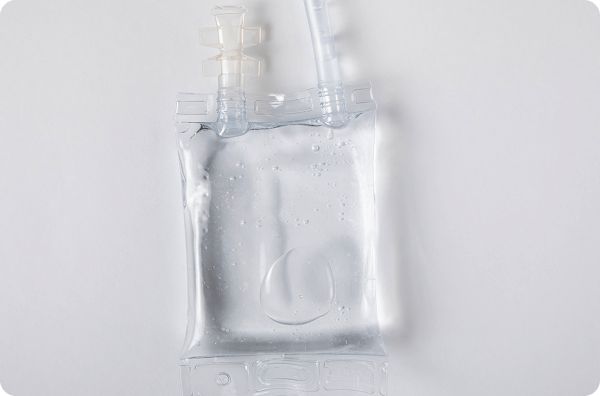
Transfer the contents of the calculated number of Thymoglobulin vials into the bag of infusion solution (saline or dextrose). Mix solution by inverting bag only once or twice
- Recommended volume: 50 mL of infusion solution per vial of Thymoglobulin (total volume usually between 50 to 500 mL)1
Thymoglobulin Administration
| Thymoglobulin should be administered under strict medical supervision in a hospital setting and patients should be carefully monitored during the infusion1 |
| Thymoglobulin is administered by intravenous injection and infused through a 0.22-μm filter into a high-flow vein (follow the manufacturer's instructions for the infusion administration set)1 |
| Set the flow rate to deliver the dose over a minimum of 6 hours for the first dose and over at least 4 hours for subsequent doses1 |
| Thymoglobulin is less likely to produce side effects when administered at the recommended flow rate1 |
| Severe hypersensitivity and infusion-related reactions, including fatal anaphylaxis and severe CRS, have been reported with the use of Thymoglobulin. Severe acute CRS can cause serious cardiorespiratory events and/or death.1 Close compliance with the recommended dosage and infusion time may reduce the incidence and severity of infusion-related reactions. Slowing the infusion rate may minimize the risk of infusion-related reactions. If a hypersensitivity or infusion-related reaction occurs, terminate the infusion immediately and provide supportive treatment according to clinical practice.1 |
Important Safety Information
CRS, cytokine release syndrome.
Reference:
1. Thymoglobulin [prescribing information]. Cambridge, MA: Genzyme Corporation; 2026.
![Graphic with the text ‘Thymoglobulin® [Anti‑thymocyte Globulin (Rabbit)]’ with an injection bottle icon on a green background, with kidney line patterns behind a woman, and a small disclaimer ‘Not an actual patient’.](https://pro.campus.sanofi/dam/jcr:51cea8f4-772a-44ce-9e7c-ed978ac758a1/SAN-THYMO-img-main-banner-administration-mobile.png)