Efficacy: Thymoglobulin® [Anti-thymocyte Globulin (Rabbit)] Offers Proven, Proactive Protection Against Acute Rejection in Kidney Transplant1
Thymoglobulin Demonstrated Superiority to Basiliximab in Reducing Treatment Failure1
The open-label, randomized, active-controlled trial compared the efficacy and safety of Thymoglobulin and of basiliximab in kidney transplant patients at increased risk for acute rejection or delayed graft function.1,2
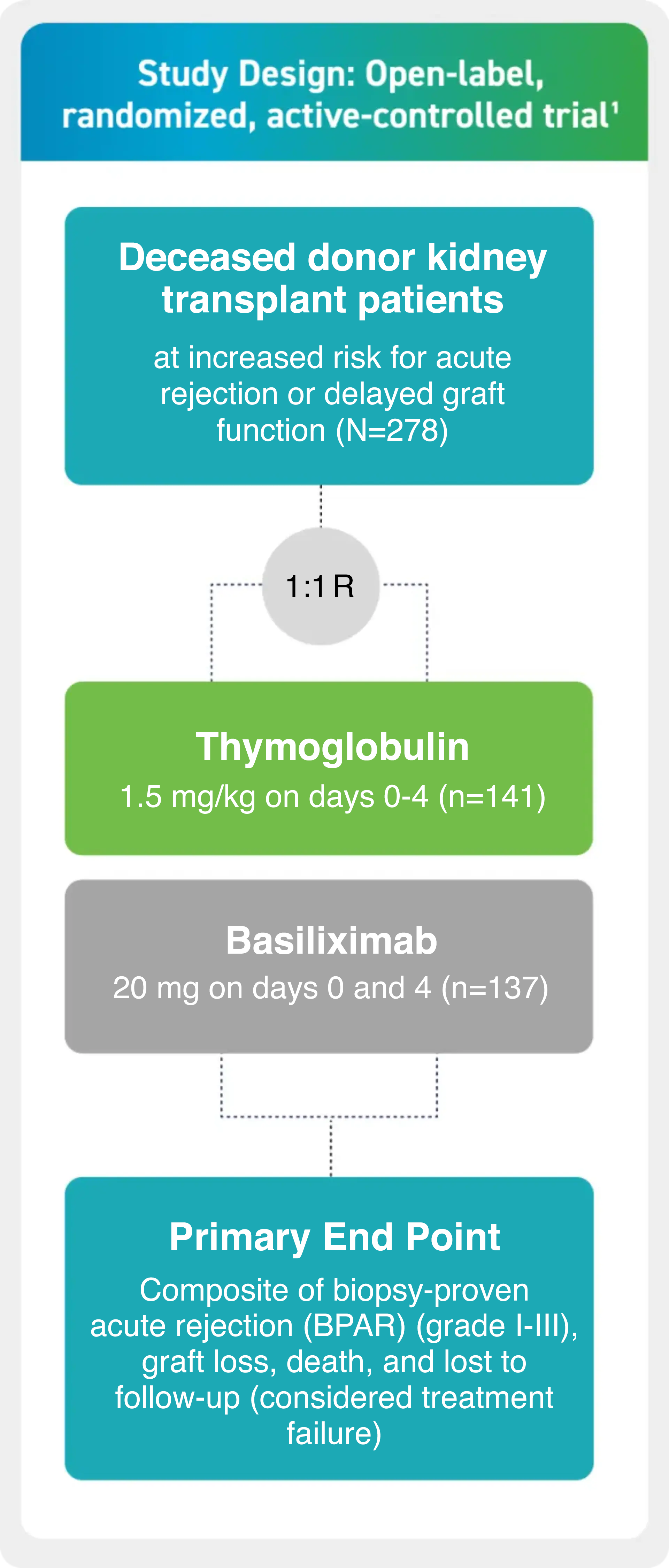
- First induction treatment was initiated prior to reperfusion of the kidney1
- All patients received triple-maintenance immunosuppression involving cyclosporine, MMF, and corticosteroids1
- Patients were followed for 12 months or until they were withdrawn from the study or lost to follow-up1
Thymoglobulin Significantly Reduced Treatment Failure Rates Compared to Basiliximab1,*
Treatment failure was based on the composite end point of BPAR, graft loss, death, or lost to follow-up within 12 months.1
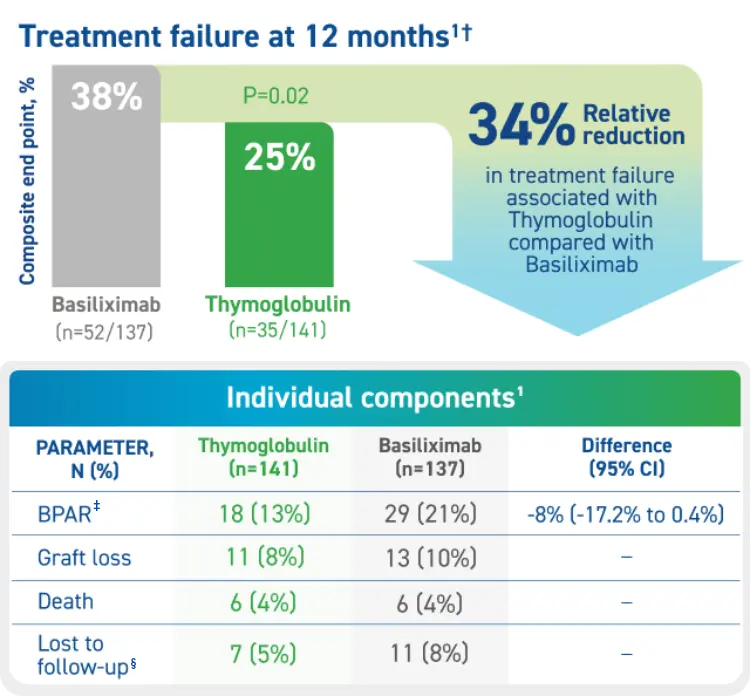
* The original primary end point of the trial published by Brennan et al was a composite of the first occurrence of BPAR, DGF, graft loss, or death.2 The FDA filing used a new composite end point, which removed DGF and included lost to follow-up, accounting for differences in the Brennan et al data compared with the Thymoglobulin label.3 The composite end point is defined as the occurrence of any of the following: BPAR (grade I-III), graft loss, death, or lost to follow-up. A patient can be counted in more than 1 category with the exception of lost to follow-up.1
†Treatment failure was defined as a composite of BPAR, graft loss, death, and lost to follow-up.1
‡BPAR is defined as the destruction of transplanted tissue or organ by the host’s immune system that has been confirmed clinically by decreased transplant organ function and biopsy.2
§Lost to follow-up was defined as not having BPAR, graft loss, or death within 12 months post transplantation, and last visit date was prior to the lower bound of 12-month window (12 months ± 30 days after transplantation).1
Thymoglobulin Demonstrated Non-inferiority to Daclizumab in Reducing Treatment Failure1
The open-label, randomized, active-controlled, investigator-sponsored trial compared BPAR incidence in high-immunological-risk renal transplant patients (n=230) receiving induction therapy with either Thymoglobulin or daclizumab.1,4

- First induction treatment was initiated prior to reperfusion of the kidney1
- All patients received triple-maintenance immunosuppression involving cyclosporine, MMF, and corticosteroids1
- Patients were followed for 12 months or until they were withdrawn from the study or lost to follow-up1
Thymoglobulin Significantly Reduced Treatment Failure Rates Compared to Daclizumab1
Treatment failure was based on the composite end point of BPAR, graft loss, death, or lost to follow-up within 12 months.1
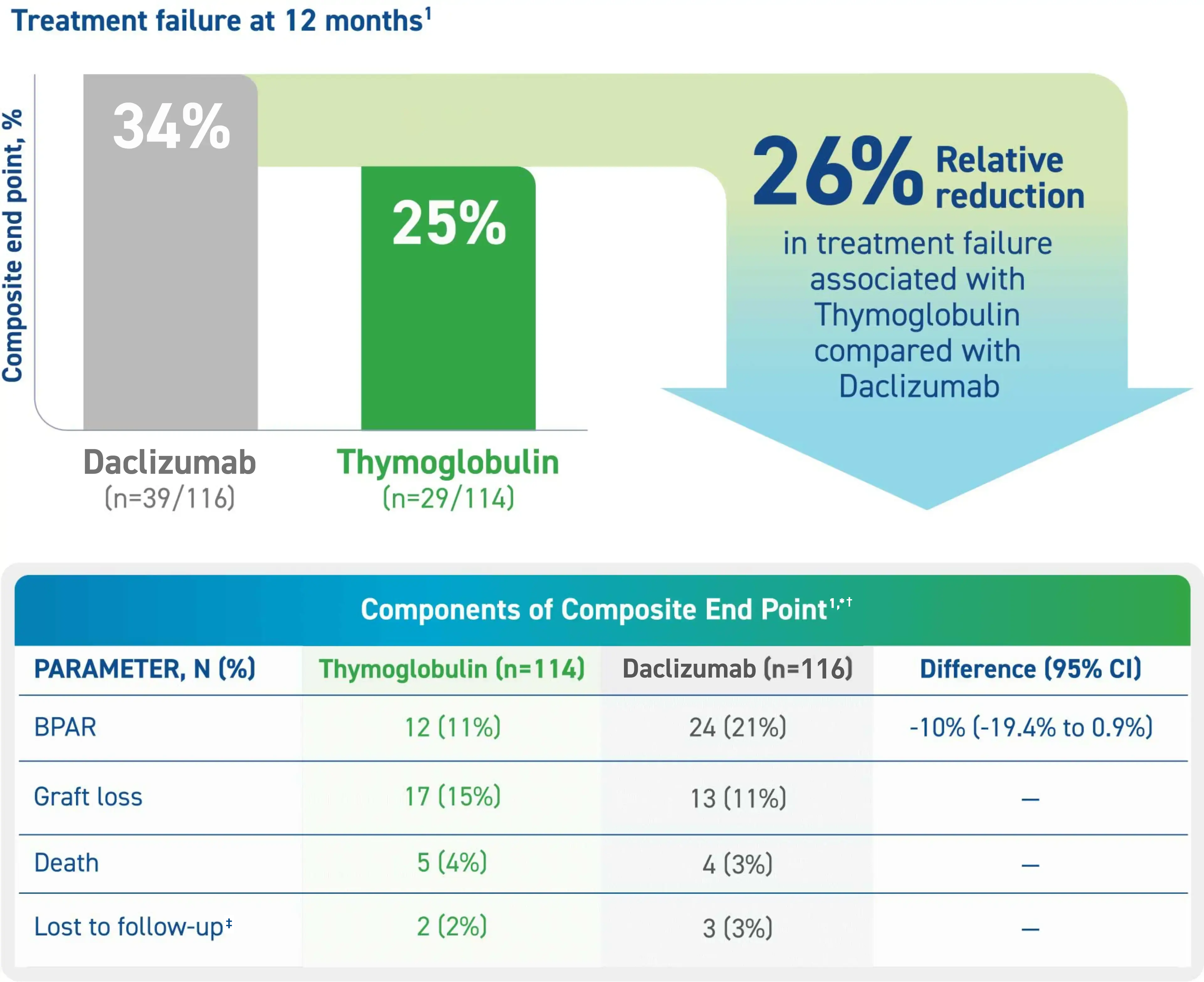
* Maximum dose of 100 mg.1
†The original primary end point of the trial published by Noël et al was BPAR within 1 year.4 The FDA filing used a common composite end point that included BPAR (grade I-III), graft loss, death, or lost to follow-up, which was included in the Thymoglobulin prescribing information. Different end points account for the differences in the treatment failure rate between the Noël publication and the Thymoglobulin prescribing information.3
‡ Lost to follow-up was defined as not having BPAR, graft loss, or death within 12 months post transplantation, and last visit date was prior to the lower bound of 12-month window (12 months ± 30 days post transplantation).1
Efficacy and Safety of Thymoglobulin: Background and Overview of Pivotal Studies
Understanding how Thymoglobulin may help in the prevention and treatment of acute rejection.
Thymoglobulin Adverse Reactions and Laboratory Abnormalities1
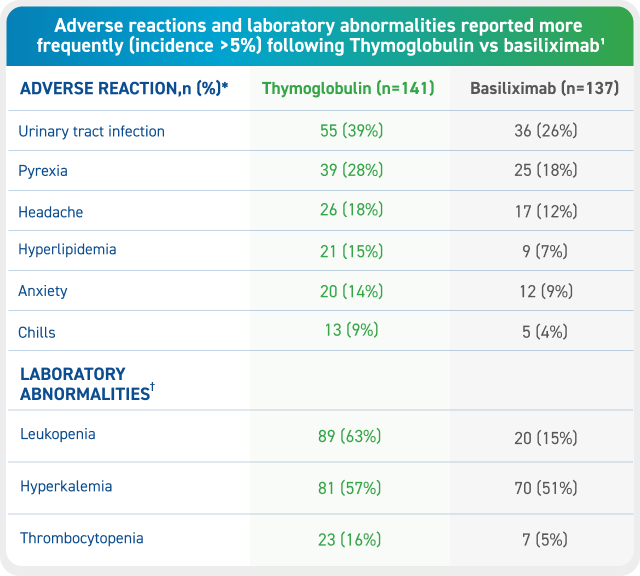
* Adverse reactions are TEAEs reported as related in at least 1 patient.1
†Leukopenia: WBC <3,000 cells /mm3; hyperkalemia: blood potassium ≥5.5 mmol /L; thrombocytopenia: platelet count <75,000 cells/mm3.1
|
Please see additional Important Safety Information below and click here for full Prescribing Information.
Important Safety Information
Abbreviations: CI, confidence interval; DGF, delayed graft function; FDA, Food and Drug Administration; MMF; Mycophenolate mofetil; TEAE, treatment-emergent adverse event; WBC, white blood cell.
References:
1. Thymoglobulin [prescribing information]. Cambridge, MA: Genzyme Corporation; 2026.
2. Brennan DC, Daller JA, Lake KD, Cibrik D, Del Castillo D; Thymoglobulin Induction Study Group. Rabbit antithymocyte globulin versus basiliximab in renal transplantation. N Engl J Med. 2006;355(19):1967-1977.
3. Data on file. sBLA Section 2.5. Sanofi Genzyme, 2015.
4. Noel C, Abramowicz D, Durand D, et al. Daclizumab versus antithymocyte globulin in high-immunological-risk renal transplant recipients. J Am Soc Nephrol. 2009;20(6):1385-1392.
![Graphic with the text 'Thymoglobulin® [Anti‑thymocyte Globulin (Rabbit)'] with document icon on a green background with kidney line patterns behind a woman and a small disclaimer 'Not an actual patient'.](https://pro.campus.sanofi/dam/jcr:3508c742-4b66-48b1-9959-99a216a95f9d/SAN-THYMO%20-%20img%20-%20main%20banner.png)