- Artikel
- Bron: Campus Sanofi
- 4 mei 2026
A Paradigm Shift in Early Detection of Autoimmune Type 1 Diabetes

Overview of current guidelines for screening and monitoring
Today, we stand at the threshold of a new era where it is possible to identify people who are developing autoimmune T1D years before symptoms appear, thus mitigating potentially life-threatening complications though timely intervention with targeted care and education.1

A Proactive Future Approach & The International Movement for Early Detection and Monitoring of Autoimmune T1D
The scientific foundation for T1D screening and monitoring has been built over decades through landmark prospective cohort studies including TEDDY, BABY-DIAB, DIPP, DiaUnion, DAISY, Fr1da, TrialNet, ELSA, and EDENT1FI.
These large-scale programs have collectively followed hundreds of thousands of individuals from birth or early childhood, generating the robust evidence base that underpins current screening and monitoring recommendations.2
A Comparative Overview of International Guidelines — spanning consensus documents from ISPAD, ADA-EASD, and EDENT1FI
The findings of large international studies for early detection of T1D have established the natural history of autoantibody development, defined the relationship between autoantibody number and progression risk, validated the biomarkers and methodological approaches that make population-level screening feasible, and determined optimal monitoring frequencies and tools for different stages of disease progression.3
This wealth of evidence has catalyzed the development of a comprehensive landscape of national guidelines, each adapting the core scientific principles to specific healthcare contexts, populations, and resource settings.4-6
These guidelines provide detailed recommendations not only for who should be screened and when, but also for how identified individuals should be staged and monitored, what tools should be used to stage and at different disease stages, when specialist referral is indicated, and how families should be educated and supported throughout the process. To date, OGTT remains the gold standard for staging when available, while alternative options such as HbA1c, CGM, fasting or random glucose are also mentioned in those guidelines.3,8
National Guidelines at a Glance — a comparative view on early detection in France, Spain, UK, Germany, and Italy
These research programs have also demonstrated that screening alone is insufficient to achieve the desired public health benefits. The reduction in DKA rates and improved outcomes are only realized when screening is coupled with systematic monitoring protocols that enable early detection of progression from stage 1 to stage 2 and ultimately to stage 3.7
International & National Recommendations for Monitoring Individuals who have tested positive to be at risk of developing T1D
The monitoring strategies developed through these studies balance the need for early detection with practical considerations of healthcare resource utilization and family burden, incorporating age-dependent protocols that reflect varying progression risks and utilizing multiple complementary tools including HbA1c measurement, glucose monitoring, oral glucose tolerance testing, and emerging technologies such as continuous glucose monitoring.8
Implementation Considerations: Feasibility, Acceptability, and Sustainability
Successful T1D screening programs depend on strong healthcare infrastructure, including trained providers and centralized laboratories. Monitoring programs must be ready from the start, as the benefits of early detection rely on active participation. Beyond clinical infrastructure, public awareness campaigns play a crucial role in reducing DKA at diagnosis. Building on these efforts, GDPR-compliant registries should standardize data across Europe to ensure long-term sustainability. Finally, equitable access for all populations is essential, with flexible options such as at-home capillary sampling to reach underserved communities.26

The Clinical Mandate for Early Detection
The current reality of autoimmune T1D diagnosis presents significant challenges that early detection can address.9
Increasingly, it is becoming common knowledge that anyone who is ultimately diagnosed with T1D will have the condition develop in 4 distinct stages,10 where the first 2 stages are asymptomatic and not detectable without proactive diagnostics efforts.
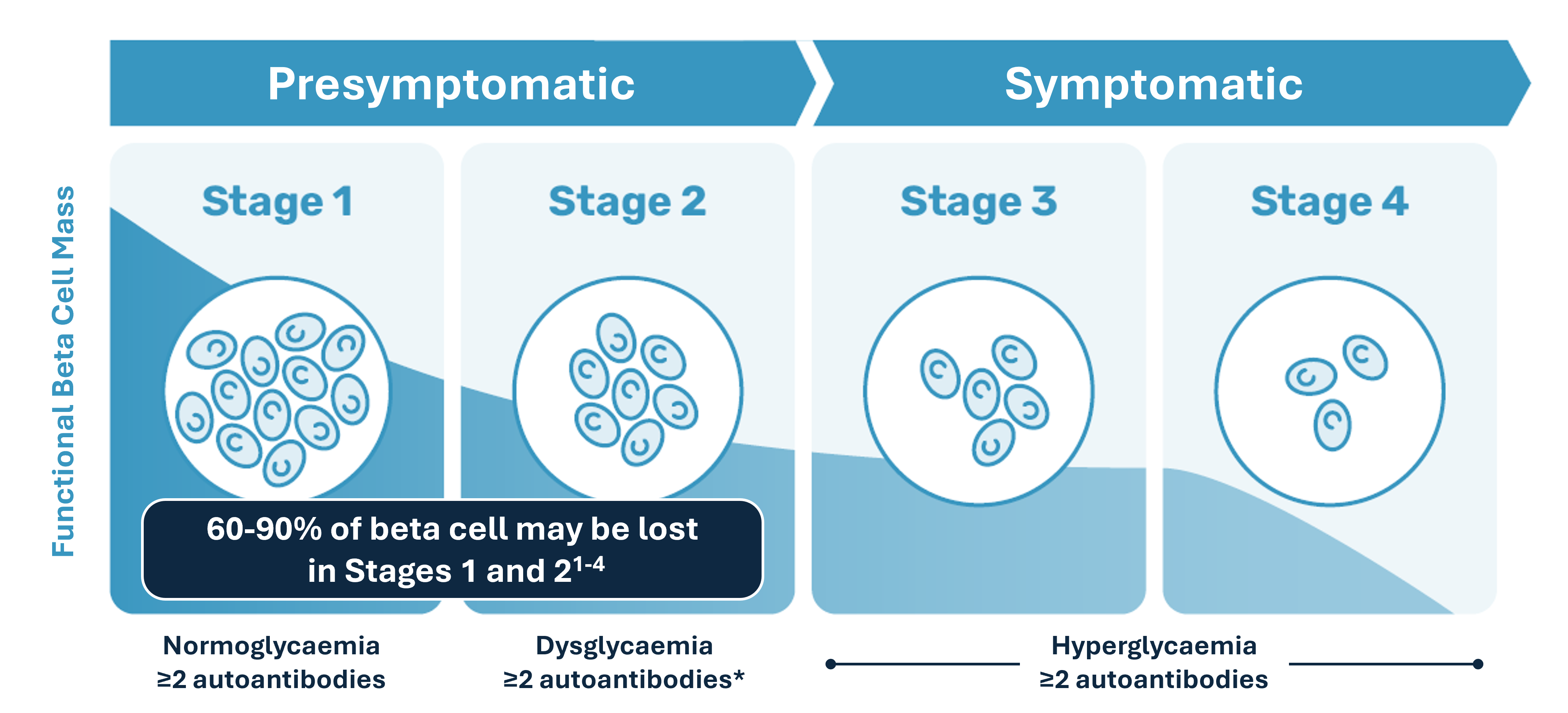
Illustration adapted from Breakthrough T1D. The stages of type 1 diabetes. Accessed March 23 2026. https://breakthrought1d.org.au/early-detection-of-type-1-diabetes/stages/
*Reversion to single autoantibody or negative status can occur in some people with previously confirmed multiple autoantibody positivity.7
As autoimmune type 1 diabetes (T1D) is driven by the progressive and irreversible loss of beta cells,11-14 it is in the interest of HCPs and people in their care to focus care plans on beta cell preservation. Here, it is interesting to note that 60-90% of functional beta cell mass may be lost in the presymptomatic stages 1 and 2.15-18
Today, most autoimmune T1D diagnoses are made when beta cell function has decreased to the point where symptoms onset. As such, a traditional approach of waiting for symptomatic disease may result in diagnoses missing critical opportunities for intervention and family preparation.19
Critically, upwards of 25-30% of children with newly diagnosed T1D presents with DKA, with this proportion rising to 30-40% in children under 5 years of age.20 DKA at diagnosis is associated with worse metabolic outcomes including higher HbA1c levels over time, more frequent hypoglycemic episodes, and increased healthcare utilization costs.21

The aim of early detection is to prevent diabetic ketoacidosis at the time of diagnosis and at the same time to determine the best time to start insulin therapy.7,9
Early detection gives those affected the opportunity to familiarize themselves with the effects that their diagnosis may have on their daily lives – before symptoms may manifest. Early detection also provides opportunities for the affected and healthcare professionals to structure education and training, which may enable gentle adjustment to life with autoimmune T1D.22-25
Sanofi is committed to providing a new understanding and perspective on Type 1 Diabetes. Join us!

-
Ziegler, A. G., Kick, K., Bonifacio, E., et al. (2020). Yield of a public health screening of children for islet autoantibodies in Bavaria, Germany. JAMA, 323(4), 339-351.
-
Sims, E. K., Besser, R. E. J., Dayan, C., et al. (2022). Screening for type 1 diabetes in the general population: a status report and perspective. Diabetes, 71(4), 610-623.
-
ISPAD Clinical Practice Consensus Guidelines 2024, Horm Res Paediatr 2024 97 (6): 529–545.
-
Mallone, R., Bismuth, E., Thivolet, C., et al. (2024). Dépistage et prise en charge du diabète de type 1 préclinique, stade 1–2. Prise de position d'experts français. Médecine des Maladies Métaboliques, 18(4), 405-432.
-
Martinez-Brocca, M. A., Bellido, V., Cardona-Hernandez, R., et al. (2025). Executive summary. Screening, staging and follow-up of type 1 diabetes in preclinical stages: Consensus of the scientific societies SED, SEEN and SEEP. Endocrinología, Diabetes y Nutrición, 72, 501639.
-
Göker et al (2025) Diabetologen Hessen eG. Vincentini, O., Pricci, F., Silano, M., et al. (2025) PLoS One, 20(8), e0328624.
-
Phillip M, Achenbach P, Addala A, et al. Diabetes Care. 2024;47(8):1276-1298.
-
Phillip M, Achenbach P, Addala A, et al. (2024). Diabetologia, 67(9), 1731-1759.
-
Insel RA, Dunne JL, Atkinson MA et al. Diabetes Care (2015) 38 1964–74.
-
American Diabetes Association. (2025). Standards of Care in Diabetes—2025. Diabetes Care 2025;48(Suppl. 1):S14–S26.
-
Herold KC, Delong T, Perdigoto AL, et al. Nat Rev Immunol. 2024;24(6):435-451.
-
O’Donovan AJ, Gorelik S, Nally LM. Front Endocrinol (Lausanne). 2024;15:1477101.
-
Ozen G, Zanfardino A, Confetto S, et al. Int J Endocrinol. 2020;2020:2630827.
-
Nagy G, Szekely TE, Somogyi A, et al. World J Diabetes. 2022;13(10):835-850.
-
Gitelman SE et al. Diabetes 2023; 72: 1289–96.
-
Wang YN et al. Front Immunol 2024; 15: 1450366.
-
Kawasaki E. Int J Mol Sci 2023; 24: 10012.
-
Breakthrough T1D. Early detection: How type 1 diabetes screening can change lives. Accessed 26 Nov. 2025. https://breakthrought1d.org.uk/resources/early-detection-how-type-1-diabetes-screening-can-change-lives/.
-
Cherubini V, et al. Diabetes Obes Metab. 2024;26:4197-202. Oron T, et al. Pediatric Diabetes. 2024. Hoffmann L, et al. BMJ Open. 2025;15(1):e088522.
-
Cherubini, V., Grimsmann, J. M., Åkesson, K., et al. (2020). Temporal trends in diabetic ketoacidosis at diagnosis of paediatric type 1 diabetes between 2006 and 2016: results from 13 countries in three continents. Diabetologia, 63(8), 1530-1541.
-
Duca, L. M., Reboussin, B. A., Pihoker, C., et al. (2019). Diabetic ketoacidosis at diagnosis of type 1 diabetes and glycemic control over time: the SEARCH for diabetes in youth study. Pediatric Diabetes, 20(2), 172-179.
-
Smith LB, et al. Pediatr Diabetes. 2018 19(5) 1025-1033
-
Joglekar MV et al. (2024) Lancet Diabetes Endocrinol. May 23 S2213-8587(24)00103-7
-
Hummel et al. (2023) Diabetologia, Vol. 66, pp. 1633–1642
-
Narendran P. Diabetologia. 2019 62(1) 24-27
-
Hussain S, Tree T, Mathieu C, et al. (2026) Diabetes Obes Metab. 28(5):3535–3556
Neem contact op

MAT-BE-2600550-v1.0-29/04/2026