The Right Dose of Thymoglobulin® [Anti-thymocyte Globulin (Rabbit)] Offers Clinically Demonstrated Protection for Your Patient's New Kidney
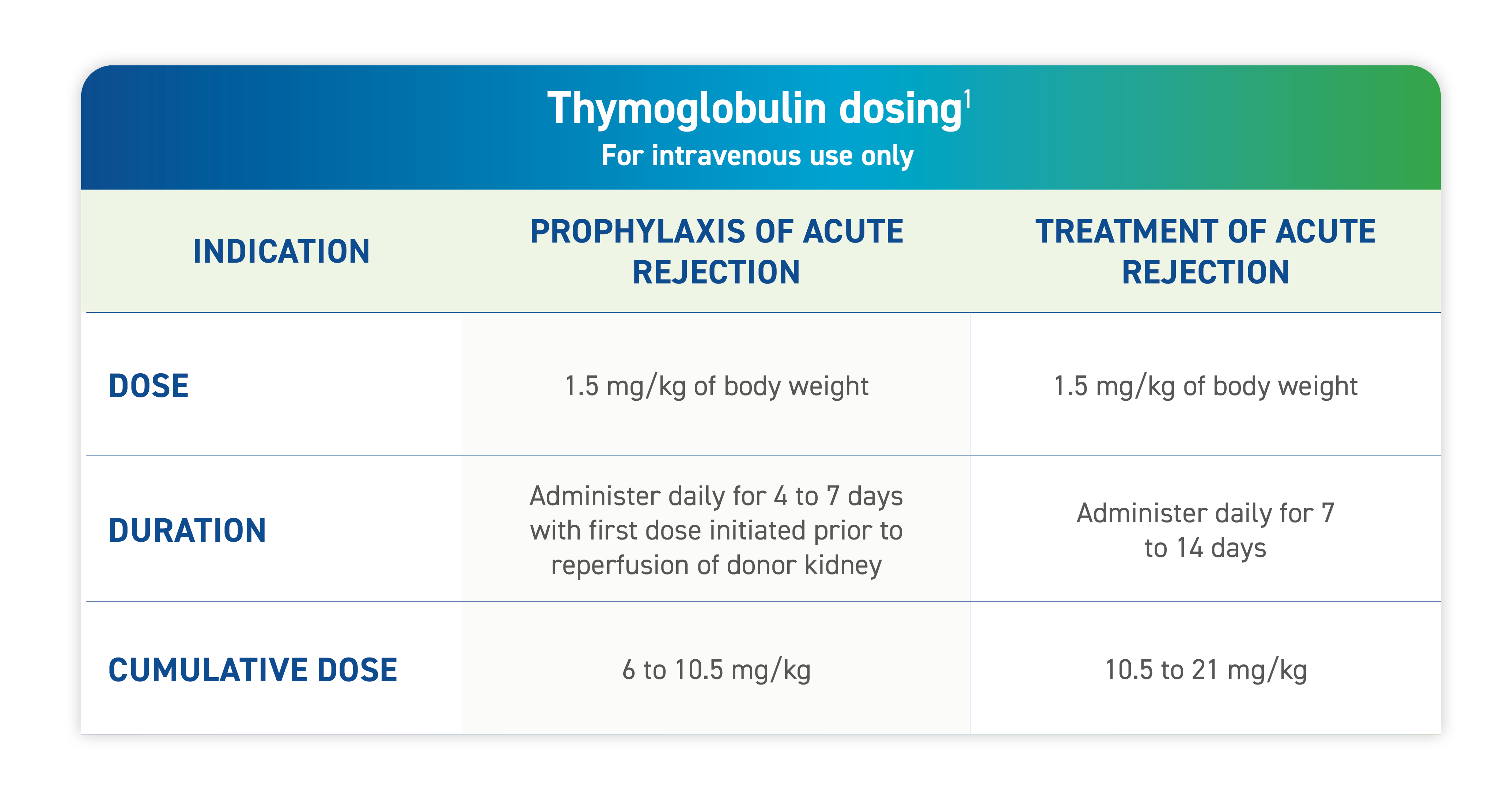
Appropriate dosing for Thymoglobulin is different from dosing for other anti-thymocyte globulin (ATG) products, as protein composition and concentrations vary depending on the source of ATG used. Physicians should therefore exercise care to ensure that the dose prescribed is appropriate for the ATG product being administered.1
Total WBC and platelet counts should be monitored to determine the need for any dose modifications1
- Reduce the Thymoglobulin dose by one-half if the WBC count is between 2,000 and 3,000 cells/mm3 or if the platelet count is between 50,000 and 75,000 cells/mm3
- Consider stopping treatment with Thymoglobulin if the WBC count falls below 2,000 cells/mm3 or if the platelet count falls below 50,000 cells/mm3
- Cytopenias including anemia, neutropenia, and thrombocytopenia have occurred with Thymoglobulin administration and require monitoring of blood counts. Adjust dose accordingly to reverse cytopenias.
- Thymoglobulin is used with concomitant immunosuppression. To prevent over-immunosuppression, physicians may wish to decrease the dose of the maintenance immunosuppression regimen during the period of Thymoglobulin use1
-
Thymoglobulin overdosage may result in leukopenia, including lymphopenia and neutropenia and/or thrombocytopenia, which can be managed with dose reductions1
Example Dose: 70 kg (154 lb) patient
The indicated dose of Thymoglobulin is 1.5 mg/kg of body weight administered daily for 4 to 7 days.
For this patient, 105 mg of Thymoglobulin would be administered per day, which would require 5 vials each day.
Over 4 to 7 days, this would equate to a cumulative dose of 420-735 mg or 20-35 vials.

Dosing Adjustments and Considerations
The immunodeficiency that results from immunosuppression predisposes patients to disorders of immune surveillance and response, including infection and malignancy. The increased risk of infection and malignancy are related more to the overall intensity of immunosuppression than to any specific agent.2,3
- To prevent over-immunosuppression, physicians may wish to decrease the dose of maintenance immunosuppression regimen during the period of Thymoglobulin use1
During Thymoglobulin therapy, monitoring total lymphocyte count may help assess the degree of T-cell depletion.1
- WBC and platelet counts should be monitored during and after therapy to assess the degree of neutropenia and thrombocytopenia, respectively1
Personalizing Thymoglobulin dose can help manage your patient’s overall immunosuppression
In all appropriate patients, the Thymoglobulin dose should be reduced to prevent over-immunosuppression1,*
Full dose of Thymoglobulin
- If WBC count is >3,000 cells/mm3
- If platelet count is >75,00 cells/mm3
Reduce dose of Thymoglobulin by half
- If WBC count is 2,000-3,000 cells/mm3
- If platelet count is 50,000-75,000 cells/mm3
Consider stopping Thymoglobulin
- If WBC count is <2,000 cells/mm3
- If platelet count is <50,000 cells/mm3
- Cytopenias including anemia, neutropenia, and thrombocytopenia have occurred with Thymoglobulin administration. Monitors blood counts after Thymoglobulin administration. Adjust dose accordingly to reverse cytopenias.1
* Monitor for adverse reactions and total WBC and platelet counts to help identify appropriate patients.1
Important Safety Information
WBC, white blood cells.
References:
1. Thymoglobulin [prescribing information]. Cambridge, MA: Genzyme Corporation; 2026.
2. Halloran PF. Immunosuppressive drugs for kidney transplantation. N Engl J Med. 2004;351(26):2715-2729.
3. Kahan BD. Individuality: the barrier to optimal immunosuppression. Nat Rev Immunol. 2003;3(10):831-838.
![Graphic with the text ‘Thymoglobulin® [Anti‑thymocyte Globulin (Rabbit)]’ with an injection bottle icon on a green background with kidney line patterns behind an elderly man and a small disclaimer ‘Not an actual patient’.](https://pro.campus.sanofi/dam/jcr:022b5e56-0f6c-42c4-b308-e557014cbf8e/SAN-THYMO%20-%20img%20-%20main%20banner-mob.png)