Irreversible lung damage may lead to chronic pulmonary conditions1,2
- In alpha-1 antitrypsin deficiency (AATD), progressive decline in lung function can lead to chronic obstructive pulmonary disease (COPD) and emphysema1,2
- This functional decline can also reduce quality of life and shorten life expectancy1-5
Patients with AATD are predisposed to developing COPD and emphysema1
- COPD exacerbations in patients with AATD can be triggered by respiratory infection, air pollution, and environmental irritants6
- The severity and frequency of COPD exacerbations can accelerate the progression of lung disease in patients with AATD7
- People with AATD-related COPD experience more emphysema, COPD exacerbations, and cirrhosis compared to those with COPD without AATD8
Emphysema is reported in 47% of Medicare patients with AATD-related COPD8
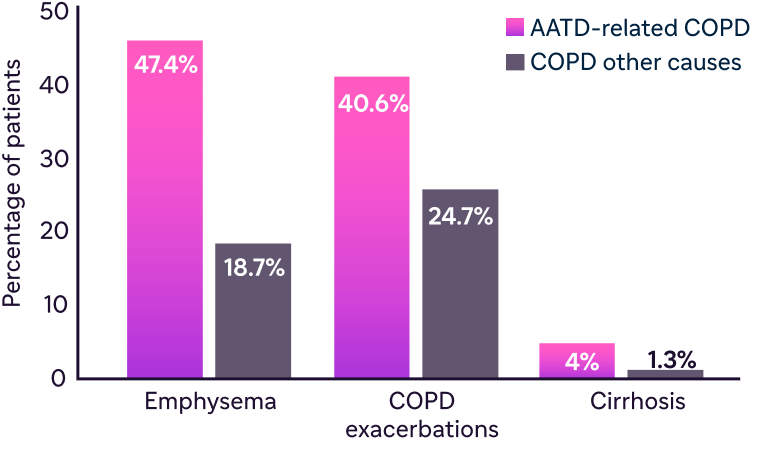
Data from Khandelwal N et al. J Health Econ Outcomes Res. 2025;12(1):66-74.
Retrospective analysis of 742 Medicare patients with AATD with emphysema, COPD exacerbations, and cirrhosis, compared with matched individuals with COPD without AATD (2015-2021).8
Severe AAT deficiency is associated with a poor prognosis with increased exacerbations and shorter lifespan5,7,9-11

Exacerbation burden is significantly higher in AATD7,9,10,12
2x
Median annualized exacerbation rates in AATD are double that observed in COPD without AATD9,10,a
61%
Of 584 patients with AATD-COPD, 61% experienced one or more severe exacerbations, including hospitalization7
14 Days
Median duration exacerbation was 14 days (interquartile range 7-21)12
aComparison of 2 different studies. Study 1: 19 patients with AATD-related COPD. Average age was 52 years. Study 2: 132 patients with moderate-to-severe COPD without AATD.
Patients with AATD have decreased life expectancy compared to the general population13
58% to 72%
of deaths in patients with severe deficiency of AAT are caused by respiratory disease such as emphysema5,11
34%
10-year survival in patients with AATD-associated COPD who received a lung transplant (1025 deceased patients)14
Patients with severe deficiency of AAT face an increased risk of early mortality, most often due to emphysema5
Mortality increases as % predicted FEV1 decreases5
Kaplan-Meier mortality analysis by % predicted FEV1
Adapted from Stoller JK et al. Chest. 2005;127(4):1196-1204.
Of the 1,129 registry enrollees, 204 subjects (18.1%) died over the course of follow-up. 7.8% (n=16/204) were never-smokers in whom emphysema was still deemed the underlying cause of death in the majority (56%, n=9/16) (National Heart, Lung, and Blood Institute Registry 1989-1992).5
-
Highest mortality is in patients with FEV1 <15%5
-
Patients with FEV1 ≥50% show the lowest mortality5
Severe AAT deficiency, defined as a serum level <11 μM.5
Emphysema is the most common cause of mortality in patients with severe deficiency of AAT5
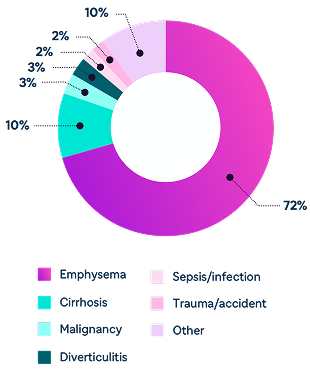
Data from Stoller JK et al. Chest. 2005;127(4):1196-1204.
Underlying causes of death among 118 patients with severe deficiency of AAT (defined as serum level <11 µmol/L). Of the 118 patients, 13.7% were never-smokers and 86.3% were ever-smokers (National Heart, Lung, and Blood Institute Registry 1989-1992).5
-
Up to 72% of deaths in patients with severe deficiency of AAT are caused by emphysema5
-
Other causes include cirrhosis, malignancy, diverticulitis, sepsis/infection, trauma/accidents, and other cases5
Progression of AATD leads to worsening COPD burden and quality of life15
The annual decline in quality of life is significantly greater for patients with AATD-associated COPD compared with AATD (no COPD) (P=0.025)3
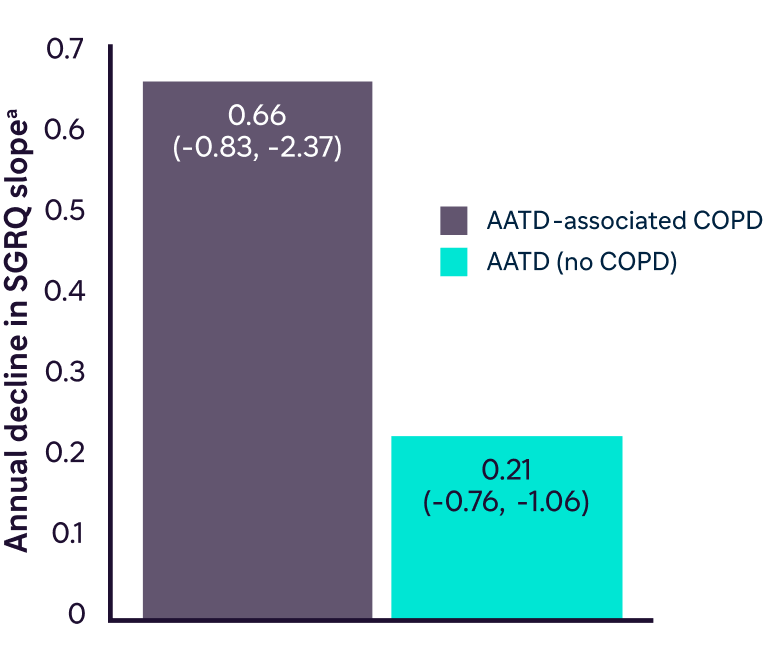
Data from Stockley RA et al. Respir Res. 2018;19(1):137.
Mean (IQR) annual decline in SGRQ total a slope in a UK retrospective database study in patients with AATD genotype ZZ (n=454).
bThe SGRQ is a commonly used measure of QoL specifically designed for patients with obstructive airway diseases.17
More frequent COPD exacerbations are associated with worsening quality of life4
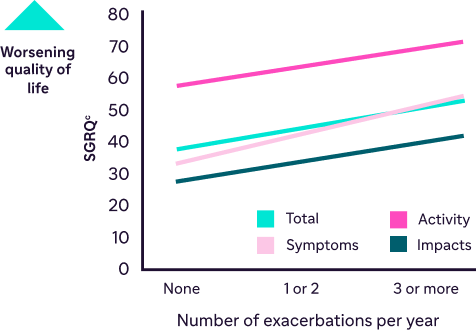
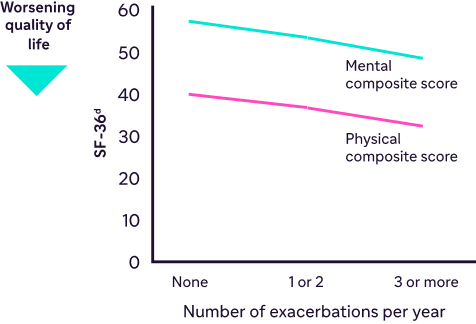
Data from Campos MA et al. Respir Med. 2009;103(10):1532-1539.
SGRQ and SF-36 scores from a noninterventional baseline follow-up of patients with AATD (N=922). Ninety-four percent (94%) of patients had genotype ZZ.4
-
Patients with AATD have more COPD exacerbations per year than patients with non-AATD COPD8
-
COPD exacerbations become more frequent as AATD progresses16
-
More frequent COPD exacerbations are associated with worsening quality of life15
cSGRQ scores range from 0 to 100, with higher scores indicating worse quality of life.17
dSF-36 scores range from 0 to 100, with lower scores indicating worse quality of life.18
AATD cannot be deduced based on age, smoking history, or severity of FEV1 decline19
FEV1 % predicted by age for 378 ZZ patients stratified by smoking status
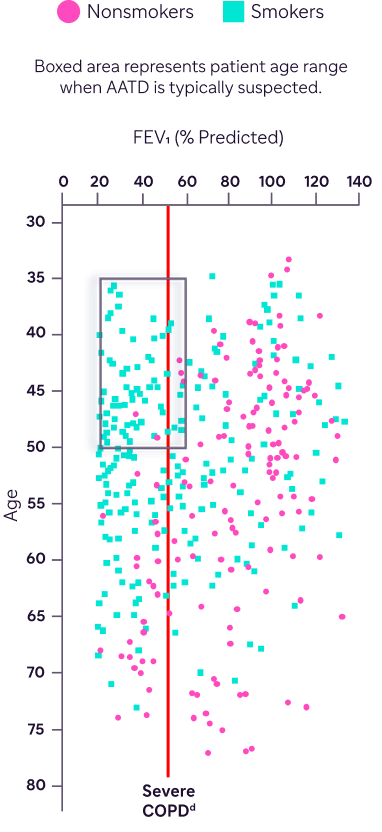
Adapted with permission from: Demeo DL et al. Thorax. 2007;62(9):806-813.
Clinical presentation alone is not enough to determine which patients with COPD may be living with less of the AAT protein.19,20
dStage III severe COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines.6

AAT=alpha-1 antitrypsin; AATD=alpha-1 antitrypsin deficiency; COPD=chronic obstructive pulmonary disease; FEV1=forced expiratory volume in 1 second; IQR=interquartile range; QoL=quality of life; SF-36=36-Item Short Form Health Survey; SGRQ=St. George’s Respiratory Questionnaire.
References: 1. Kalfopoulos M, Wetmore K, ElMallah MK. Pathophysiology of alpha-1 antitrypsin lung disease. In: Borel F, Mueller C, eds. Alpha-1 Antitrypsin Deficiency: Methods in Molecular Biology. Humana Press; 2017:9-20. 2. Meseeha M, Sankari A, Attia M. Alpha-1 antitrypsin deficiency. In: StatPearls [Internet]. StatPearls Publishing; August 17, 2024. Accessed April 14, 2026. https://www.ncbi.nlm.nih.gov/books/NBK442030/ 3. Stockley RA, Edgar RG, Starkey S, Turner AM. Health status decline in α-1 antitrypsin deficiency: a feasible outcome for disease modifying therapies? Resp Res. 2018;19(1):137. 4. Campos MA, Alazemi S, Zhang G, et al. Exacerbations in subjects with alpha-1 antitrypsin deficiency receiving augmentation therapy. Respir Med. 2009:103(10)1532-1539. 5. Stoller JK, Tomashefski J Jr, Crystal RG, et al. Mortality in individuals with severe deficiency of alpha1-antitrypsin: findings from the National Heart, Lung, and Blood Institute Registry. Chest. 2005;127(4):1196-1204. 6. Global Initiative for Chronic Obstructive Lung Disease. GOLD Report. Accessed April 14, 2026. https://goldcopd.org 7. Hiller AM, Piitulainen E, Jehpsson L, Tanash H. Decline in FEV1 and hospitalized exacerbation in individuals with severe alpha-1 antitrypsin deficiency. Int J Chron Obstruct Pulm Dis. 2019;14:1075-1083. 8. Khandelwal N, Hinson J, Nguyen T, et al. Clinical and economic outcomes in patients with alpha-1 antitrypsin deficiency in a US Medicare advantage population. J Health Econ Outcomes Res. 2025;12(1):66-74. 9. Vijayasaratha K, Stockley RA. Reported and unreported exacerbations of COPD: analysis by diary cards. Chest. 2008;133(1):34-41. 10. Donaldson GC, Seemungal TA, Patel IS, Lloyd-Owen SJ, Wilkinson TM, Wedzicha JA. Longitudinal changes in the nature, severity and frequency of COPD exacerbations. Eur Respir J. 2003;22(6):931-936. doi:10.1183/09031936.03.00038303 11. Tanash HA, Nilsson PM, Nilsson JA, Piitulainen E. Survival in severe alpha-1-antitrypsin deficiency (PiZZ). Respir Res. 2010;11(1):44. doi:10.1186/1465-9921-11-44 12. Needham M, Stockley RA. Exacerbations in α1-antitrypsin deficiency. Eur Respir J. 2005;25(6):992-1000. 13. Wahlin S, Widman L, Hagström H. Epidemiology and outcomes of alpha-1 antitrypsin deficiency in Sweden 2002-2020: a population-based cohort study of 2286 individuals. J Intern Med. 2025;297(3):300-311. 14. Riley L, Lascano J. Clinical outcomes and survival following lung transplantation in patients with Alpha-1 antitrypsin deficiency. Respir Med. 2020;172:106145. doi:10.1016/j.rmed.2020.106145 15. Choate R, Holm KE, Sandhaus RA, Mannino DM, Strange C. Characteristics associated with SF-36 in alpha-1 antitrypsin deficiency-associated COPD: a cross-sectional analysis. BMC Pulm Med. 2024;24(1):138. doi:10.1186/s12890-024-02953-7 16. Miravitlles M, Herepath M, Priyendu A, et al. Disease burden associated with alpha-1 antitrypsin deficiency: systematic and structured literature reviews. Eur Respir Rev. 2022;31(163):210262. doi:10.1183/16000617.0262-2021 17. Jones P. St. George’s Respiratory Questionnaire Manual. St. George’s, University of London; March 2022. Accessed March 11, 2026. https//www.citystgeorges.ac.uk/__data/assets/pdf_file/0009/899631/SGRQ-Manual-March-2022.pdf 18. Ware JE, Snow KK, Kosinski M, Gandek B. SF-36 Health Survey Manual and Interpretation Guide. The Health Institute, New England Medical Center; 1993. Accessed April 16, 2026. https://www.researchgate.net/profile/John-Ware-6/publication/313050850_SF-36_Health_Survey_Manual_Interpretation_Guide/links/594a5b83aca2723195de5c3d/SF-36-Health-Survey-Manual-Interpretation-Guide.pdf 19. Demeo DL, Sandhaus RA, Barker AF, et al. Determinants of airflow obstruction in severe alpha-1-antitrypsin deficiency. Thorax. 2007;62(9):806-813. 20. Molloy K, Hersh CP, Morris VB, et al. Clarification of the risk of chronic obstructive pulmonary disease in α1-antitrypsin deficiency PiMZ heterozygotes. Am J Respir Crit Care Med. 2014;189(4):419-427. 21. American Thoracic Society/European Respiratory Society Statement: Standards for the Diagnosis and Management of Individuals with Alpha-1 Antitrypsin Deficiency. Am J Respir Crit Care Med. 2003;168(7):818-900. 22. Sandhaus RA, Turino G, Brantly ML, et al. The diagnosis and management of alpha-1 antitrypsin deficiency in the adult. Chron Obstr Pulm Dis. 2016;3(3):668-682. 23. Abboud RT, Nelson TN, Jung B, Mattman A. Alpha1-antitrypsin deficiency: a clinical-genetic overview. Appl Clin Genet. 2011;4:55-65. 24. Mazzuca C, Vitiello L, Travaglini S, et al. Immunological and homeostatic pathways of alpha-1 antitrypsin: a new therapeutic potential. Front Immunol. 2024;15:1443297. doi:10.3389/fimmu.2024.1443297 25. Feitosa PHR, de Oliveira Castellano MVC, da Costa CH, et al. Recommendations for the diagnosis and treatment of alpha-1 antitrypsin deficiency. J Bras Pneumol. 2024;50(5):e20240235. doi:10.36416/1806-3756/e20240235

