Genotypes and resultant AAT levels drive increased risk of irreversible lung damage1,2
- In individuals without alpha-1 antitrypsin deficiency (AATD) (MM genotype), normal alpha-1 antitrypsin (AAT) levels are between 20 to 53 µM3
- Pathogenic variants of the SERPINA1 gene reduce circulating AAT protein4
- AAT levels vary across genotypes and span a spectrum of severity3,5
- Reduced AAT levels increase vulnerability to faster progression such as loss of lung function over time1,6
- Behavioral, environmental, and physiologic factors can further influence this risk6-9
AATD risk assessment begins with defining the underlying genetics1,5
AATD is caused by pathogenic variants of SERPINA1, the gene that encodes the AAT protein. Key alleles include1,4:
M allele
(Baseline, normal function)1,5
- Produces normal, functional AAT
- Not associated with increased lung or liver risk
- Most common allele in the general population
S allele
(Mild to moderate deficiency)1,5
- Produces less AAT than M, but significantly more than Z
- Polymerizes slowly, so liver involvement is uncommon
- Lung risk increases when paired with Z (eg, SZ genotype)
Z allele
(Highest clinical impact)1,5
- Severely reduced circulating AAT levels
- Misfolded AAT accumulates in hepatocytes, contributing to liver injury
- ZZ individuals have the highest risk for both lung disease and liver complications
- Most common disease-causing variant
Q0/Null allele
(No AAT production)1,5
- No detectable AAT in circulation
- High lung risk due to complete absence of AAT
- Not associated with liver disease because no misfolded protein accumulates
The risks of AATD fall on a spectrum of severity3,5
Risk Spectrum
|
MM |
MZ |
SZ |
ZZ | |
|
Normal5 |
Mild to moderate AAT deficiency5 |
Moderate AAT deficiency3,5 |
Severe AAT deficiency5 | |
|
Plasma AAT Levels |
20-53 µM3 |
15-42 µM3 |
10-23 µM3 |
3.4-7 µM3 |
|
Mechanism |
|
|
|
|
|
Implication |
|
|
|
|
|
Key Takeaway |
|
|
|
|
The lower AAT levels are, the higher the risk of AATD-associated emphysema3
Historically, protective AAT levels have been benchmarked at 11 µM. However, the normal range is 20-53 µM.3,11
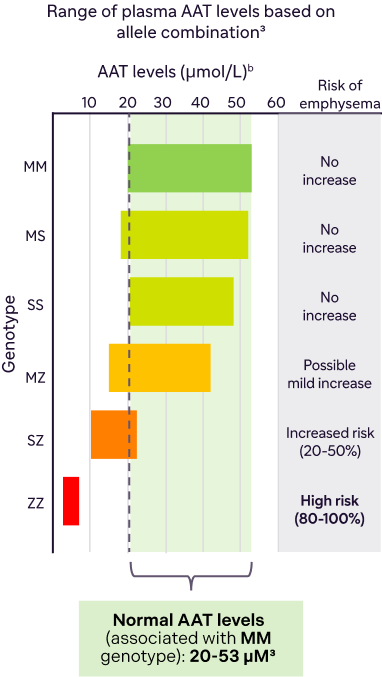
Data from Mulkareddy V et al. Am J Med Sci. 2024;368(1):1-8.
aAAT levels may also be measured in mg/dL.
Risk is multifactorial and can be influenced by lifestyle and environmental factors.7
Genotype is a determinant of risk, and all intermediate and severe genotypes carry potential risk.3
Besides genotype, other factors can contribute to risk in AATD1,6-8
Additional risk factors include6-8:
Behavioral
Smoking
Environmental
Air pollution, occupational inhalants

Pathophysiologic
Respiratory infections
Smoking can significantly accelerate the rate of lung decline in patients with AATD9
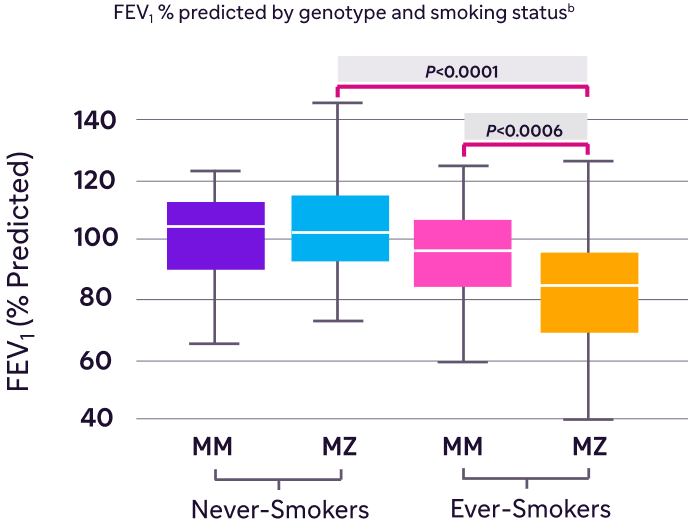
Figure adapted from Molloy K et al. Am J Respir Crit Care Med. 2014;189(4):419-427.
bIncluded 99 individuals with MM genotype and 89 individuals with MZ genotype.
In MZ individuals, FEV1 was significantly lower in ever-smokers compared with never-smokers9
- Never-smoker was defined as less than 20 packs of cigarettes, with 12 oz of tobacco in a lifetime, or less than 1 cigarette a day for 1 year9
- Approximately 2% to 3% of the Caucasian population is born with the MZ genotype1
AAT=alpha-1 antitrypsin; AATD=alpha-1 antitrypsin deficiency; COPD=chronic obstructive pulmonary disease; FEV1=forced expiratory volume in 1 second.
References: 1. American Thoracic Society/European Respiratory Society Statement: Standards for the Diagnosis and Management of Individuals with Alpha-1 Antitrypsin Deficiency. Am J Respir Crit Care Med. 2003;168(7):818-900. 2. Meseeha M, Sankari A, Attia M. Alpha-1 antitrypsin deficiency. In: StatPearls [Internet]. StatPearls Publishing; August 17, 2024. Accessed April 14, 2026. https://www.ncbi.nlm.nih.gov/books/NBK442030/ 3. Mulkareddy V, Roman J. Pulmonary manifestations of alpha 1 antitrypsin deficiency. Am J Med Sci. 2024;368(1):1-8. 4. Cazzola M, Stolz D, Rogliani P, Matera MG. α1-Antitrypsin deficiency and chronic respiratory disorders. Eur Respir Rev. 2020;29(155):190073. doi:10.1183/16000617.0073-2019 5. Feitosa PHR, de Oliveira Castellano MVC, da Costa CH, et al. Recommendations for the diagnosis and treatment of alpha-1 antitrypsin deficiency. J Bras Pneumol. 2024;50(5):e20240235. doi:10.36416/1806-3756/e20240235 6. Wang T, Shuai P, Wang Q, et al. α‑1 Antitrypsin is a potential target of inflammation and immunomodulation (Review). Mol Med Rep. 2025;31(4):107. doi:10.3892/mmr.2025.13472 7. Torres-Durán M, Lopez-Campos JL, Barrecheguren M, et al. Alpha-1 antitrypsin deficiency: outstanding questions and future directions. Orphanet J Rare Dis. 2018;13(1):114. doi:10.1186/s13023-018-0856-9 8. Kokturk N et al. Lung inflammation in alpha‑1‑antitrypsin deficient individuals with normal lung function. Respir Res. 2023;24(1):40. 9. Molloy K, Hersh CP, Morris VB, et al. Clarification of the risk of chronic obstructive pulmonary disease in α1-antitrypsin deficiency PiMZ heterozygotes. Am J Respir Crit Care Med. 2014;189(4):419-427. 10. McElvaney GN, Sandhaus RA, Miravitlles M, et al. Clinical considerations in individuals with α1-antitrypsin PI*SZ genotype. Eur Respir J. 2020;55(6):1902410. doi: 10.1183/13993003.02410-2019 11. Stoller JK, Aboussouan LS. A review of α1-antitrypsin deficiency. Am J Respir Crit Care Med. 2012;185(3):246-259. 12. Turino GM, Barker AF, Brantly ML, et al. Clinical features of individuals with PI*SZ phenotype of alpha 1-antitrypsin deficiency. Alpha 1-Antitrypsin Deficiency Registry Study Group. Am J Respir Crit Care Med. 1996;154(6)(pt 1):1718-1725. 13. Vijayasaratha K, Stockley RA. Reported and unreported exacerbations of COPD: analysis by diary cards. Chest. 2008;133(1):34-41. 14. Donaldson GC, Seemungal TA, Patel IS, Lloyd-Owen SJ, Wilkinson TM, Wedzicha JA. Longitudinal changes in the nature, severity and frequency of COPD exacerbations. Eur Respir J. 2003;22(6):931-936. doi:10.1183/09031936.03.00038303 15. Wahlin S, Widman L, Hagström H. Epidemiology and outcomes of alpha-1 antitrypsin deficiency in Sweden 2002-2020: a population-based cohort study of 2286 individuals. J Intern Med. 2025;297(3):300-311. 16. Stoller JK, Tomashefski J Jr, Crystal RG, et al. Mortality in individuals with severe deficiency of alpha1-antitrypsin: findings from the National Heart, Lung, and Blood Institute Registry. Chest. 2005;127(4):1196-1204. 17. Global Initiative for Chronic Obstructive Lung Disease. GOLD Report. Accessed April 14, 2026. https://goldcopd.org 18. Sandhaus RA, Turino G, Brantly ML, et al. The diagnosis and management of alpha-1 antitrypsin deficiency in the adult. Chron Obstr Pulm Dis. 2016;3(3):668-682.

