What is the mechanism of disease behind AATD?
- Alpha-1 antitrypsin (AAT) is a protein that protects against protease-induced damage1
- AAT is primarily synthesized in the liver and diffuses into the lungs where it keeps elastase activity in check1,2
- Below-normal levels of AAT result in alpha-1 antitrypsin deficiency (AATD)3,4
- This inherited condition puts the lungs and other areas of the body at risk of irreversible damage4,5
AAT protects lung tissue from protease-induced damage1
AAT plays a key role in protecting the lungs by1:
- Inhibiting neutrophil elastase and dampening pro-inflammatory cytokine production
- Reducing cellular damage from reactive oxygen species (ROS)
- Supporting immunomodulatory pathways that help protect tissues from unchecked inflammatory activity

Smoking, air pollution, and dust exposure are also contributing factors to increased risk of accelerated lung disease. Pathophysiologic factors such as respiratory infection may also play a role.6-8


AAT is primarily synthesized in the liver and diffuses into the lungs where it keeps elastase activity in check1
8%
of newborns with severe AAT deficiency present with severe liver disease9,a
81%
of patients with severe AAT deficiency self-report obstructive lung disease11,b
aStudy that evaluated 200,000 newborns in Sweden identified the ZZ genotype in 127, of whom 73% had prolonged jaundice and 8% had severe liver disease.9
bSelf-administered questionnaire with 1,953 responders. Smoking history was not queried. Severe deficiency of AAT (SZ, ZZ, and Z/null genotypes) was present in 92.7% of the survey participants (n=1,810).11
Within the lung12:
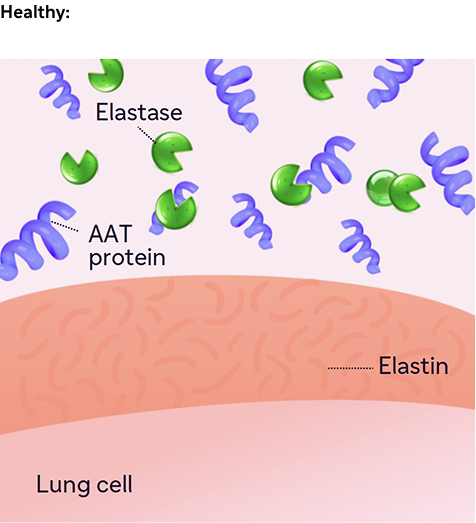
AAT protein inhibits neutrophil elastase, thus preventing elastin degradation.
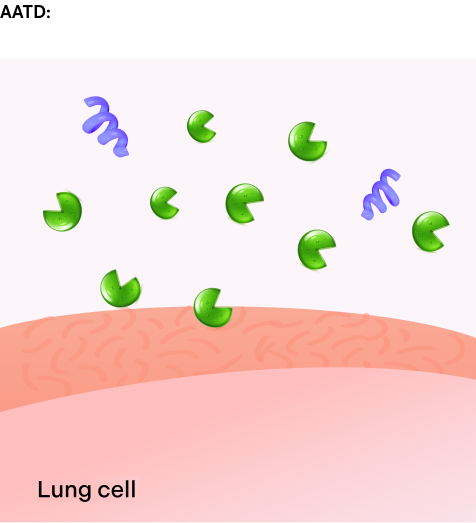
Without sufficient levels of AAT protein, elastase activity is unchecked, causing damage to the lung tissue and restricting airflow.
Deficient AAT can result in pro-inflammatory signaling and ROS-mediated damage1,13
Inflammatory signaling and immune cell differentiation1,13
- In physiological conditions, AAT protein dampens pro-inflammatory cytokine production, inhibiting neutrophil activity, and modulating immune cell differentiation and proliferation
- In AATD, low circulating AAT protein leads to a pro-inflammatory state in the lungs, resulting in structural lung-cell damage and apoptosis
Oxidation1,4,14
- Oxidation of AAT protein by free ROS prevents cellular damage and reduces further inflammatory signaling
- However, oxidation of AAT protein renders it inactive, which leads to a decrease in AAT’s antielastase activity, which is problematic in patients with already low levels of circulating AAT
AAT=alpha-1 antitrypsin; AATD=alpha-1 antitrypsin deficiency; COPD=chronic obstructive pulmonary disease; ROS=reactive oxygen species.
References: 1. Mazzuca C, Vitiello L, Travaglini S, et al. Immunological and homeostatic pathways of alpha-1 antitrypsin: a new therapeutic potential. Front Immunol. 2024;15:1443297. doi:10.3389/fimmu.2024.1443297 2. de Serres FJ et al. J Intern Med. 2014;276(4):311-335. 3. American Thoracic Society; European Respiratory Society. American Thoracic Society/European Respiratory Society Statement: Standards for the Diagnosis and Management of Individuals with Alpha-1 Antitrypsin Deficiency. Am J Respir Crit Care Med. 2003;168(7):818-900. 4. Kalfopoulos M, Wetmore K, ElMallah MK. Pathophysiology of alpha-1 antitrypsin lung disease. In: Borel F, Mueller C, eds. Alpha-1 Antitrypsin Deficiency: Methods in Molecular Biology. Humana Press; 2017:9-20 5. Meseeha M, Sankari A, Attia M. Alpha-1 antitrypsin deficiency. In: StatPearls [Internet]. StatPearls Publishing; August 17, 2024. Accessed March 4, 2026. https://www.ncbi.nlm.nih.gov/books/NBK442030/ 6. Torres-Durán M, Lopez-Campos JL, Barrecheguren M, et al. Alpha-1 antitrypsin deficiency: outstanding questions and future directions. Orphanet J Rare Dis. 2018;13(1):114. doi:10.1186/s13023-018-0856-9 7. Wang T, Shuai P, Wang Q, et al. α‑1 antitrypsin is a potential target of inflammation and immunomodulation. Mol Med Rep. 2025;31(4):107. doi:10.3892/mmr.2025.13472 8. Kokturk N et al. Lung Inflammation in alpha‑1‑antitrypsin deficient individuals with normal lung function. Respir Res. 2023;24(1):40. 9. Feitosa PHR, de Oliveira Castellano MVC, da Costa CH, et al. Recommendations for the diagnosis and treatment of alpha-1 antitrypsin deficiency. J Bras Pneumol. 2024;50(5):e20240235. doi:10.36416/1806-3756/e20240235 10. Cazzola M, Stolz D, Rogliani P, Matera MG. α1-Antitrypsin deficiency and chronic respiratory disorders. Eur Respir Rev. 2020;29(155):190073. doi:10.1183/16000617.0073-2019 11. Strange C, Stoller JK, Sandhaus RA, Dickson R, Turino G. Results of a survey of patients with alpha-1 antitrypsin deficiency. Respiration. 2006:73(2):185-190. doi:10.1159/000088061 12. University of Utah Health. Alpha-1 antitrypsin deficiency. Accessed April 16, 2026. https://learn.genetics.utah.edu/content/genetics/alpha1/ 13. Toumpanakis D, Usmani OS. Small airways disease in patients with alpha-1 antitrypsin deficiency. Respir Med. 2023;211:107222. doi:10.1016/j.rmed.2023.107222 14. Magallón M, Pastor S, Carrión AE, et al. Oxidative stress and endoplasmic reticulum stress in rare respiratory diseases. J Clin Med. 2021;10(6):1268. doi:10.3390/jcm10061268 15. Vijayasaratha K, Stockley RA. Reported and unreported exacerbations of COPD: analysis by diary cards. Chest. 2008;133(1):34-41. doi:10.1378/chest.07-1692 16. Donaldson GC, Seemungal TA, Patel IS, Lloyd-Owen SJ, Wilkinson TM, Wedzicha JA. Longitudinal changes in the nature, severity and frequency of COPD exacerbations. Eur Respir J. 2003;22(6):931-936. doi:10.1183/09031936.03.00038303 17. Wahlin S, Widman L, Hagström H. Epidemiology and outcomes of alpha-1 antitrypsin deficiency in Sweden 2002-2020: a population-based cohort study of 2286 individuals. J Intern Med. 2025;297(3):300-311. doi:10.1111/joim.20058 18. Stoller JK, Tomashefski J Jr, Crystal RG, et al. Mortality in individuals with severe deficiency of alpha-1 antitrypsin: findings from the National Heart, Lung, and Blood Institute Registry. Chest. 2005;127(4):1196-1204. 19. Global Initiative for Chronic Obstructive Lung Disease (GOLD). GOLD Report. Accessed February 3, 2026. https://goldcopd.org 20. Sandhaus RA, Turino G, Brantly ML, et al. The diagnosis and management of alpha-1 antitrypsin deficiency in the adult. Chron Obstr Pulm Dis. 2016;3(3):668-682.

